Use cases
Temperature compliance for every environment in your pharma supply chain. From cold storage rooms to cargo flights, see how Eupry helps you stay compliant and in control.

Autoclaves
Autoclave validation without the box. Wireless loggers that survive steam sterilization, with real-time monitoring and automated compliance documentation.
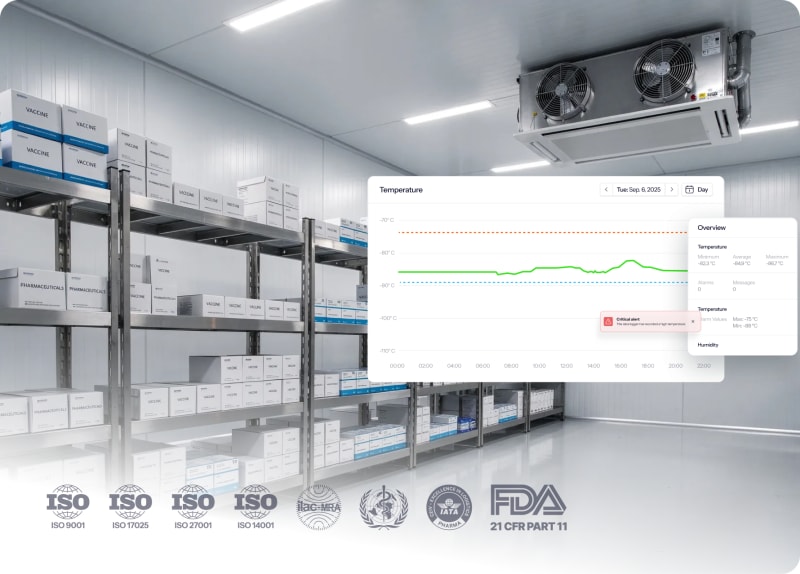
Cold storage rooms
Complete visibility across multiple cold rooms and sites. Standardized processes and audit-ready documentation for FDA, EMA, and more.

Fridges and freezers
24/7 visibility for vaccines, biologics, and temperature-sensitive samples. Instant alerts, built-in redundancy, and audit-ready documentation for FDA, EMA, and more.

Incubators and climate chambers
You run stability studies or cell cultures that require precise and stable temperature and humidity control. You need reliable monitoring that catches problems immediately, not during the next check.

Laboratories
You run a lab with multiple assets requiring environmental control with more than just temperature. You need one system that monitors everything and handles compliance automatically.

Pharma cargo flights
Complete temperature records from origin to destination. Data syncs automatically when shipments land. In-flight alerts enable pilots to take corrective action.

Reefers & Active ULDs
You use temperature controlled containers for pharmaceutical transport and need independent temperature verification. You need ruggedized loggers that sync automatically at destination.

Ultra low freezers (ULTs) and cryos
24/7 visibility for samples stored from -80°C to -196°C. Instant alerts, built-in redundancy, and audit-ready documentation for FDA, EMA, and more.
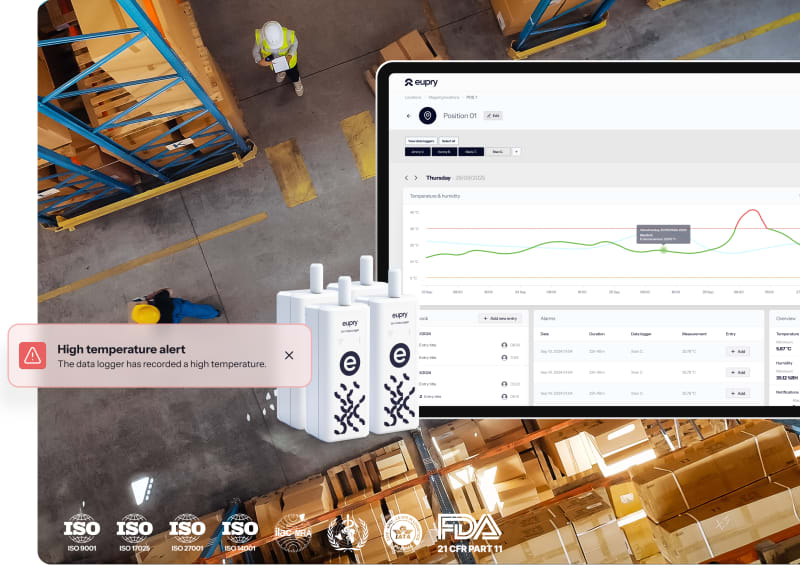
Warehouses
GDP compliance documentation that impresses clients. Validation that does not disrupt operations. One provider for monitoring, mapping, and calibration.
Do not see your environment listed?
Our loggers and platform support a wide range of GxP-regulated applications beyond what is shown here. Get in touch here to discuss your specific requirements.