Autoclave monitoring for pharmaceutical manufacturing
Autoclave validation without the box. Wireless loggers that survive steam sterilization, with real-time monitoring and automated compliance documentation.

You find out too late
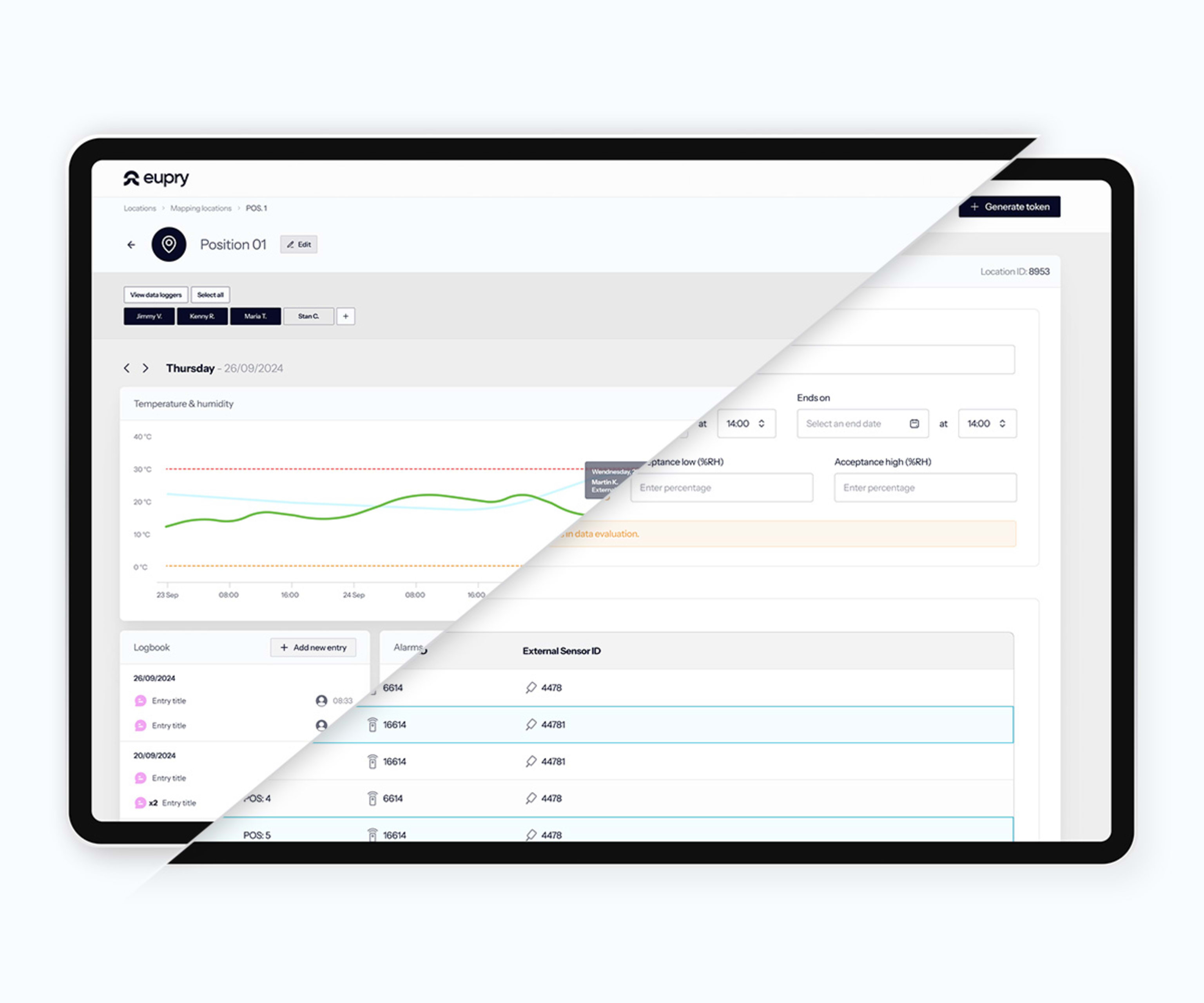
Incubators and climate chambers contain valuable research that cannot tolerate excursions. Manual monitoring misses problems between checks. Traditional systems are clunky and require IT support. When something goes wrong at 2 AM, you find out at 8 AM. When you assess the damage and the impact, the traditional systems do not provide you with the data and insights you need to make a proper assessment.

Alerts the moment it matters
Eupry monitors temperature and humidity continuously inside your incubators and chambers. Real-time alerts via SMS and email if conditions drift. Compliant records, audit reports and calibration certificates in 3 clicks. Calibration included, happens on-site without any downtime.


“Eupry was very professional and informative throughout the process, providing us with high-quality service (...) It's undoubtedly a well-put partnership.”
Joakim Sund
Quality Assurance Specialist

“Eupry was instrumental in helping us establish a GDP-compliant operation ready for the standards of pharmaceutical logistics. Their flexibility, expertise, and collaborative approach were exactly what we needed to navigate the requirements of the industry and develop a foundation that worked for us."

Michael O'Brien
President

“All of the loggers have proven their worth and saved precious samples on several occasions when freezers broke or were accidentally left open.“

Anja Pomowski
Senior Scientist Antibody Production at Antikor
Relevant products

Download our Product Catalog
Get an overview of how Eupry’s products can help with automatic monitoring and mapping of temperatures, humidity, CO2 and diff. pressure.
Want to know more?
Discover how Eupry's automated compliance (monitoring, calibration, and validation) solutions work today.