ULT and cryo monitoring for pharmaceutical storage
24/7 visibility for samples stored from -80°C to -196°C. Instant alerts, built-in redundancy, and audit-ready documentation for FDA, EMA, and more.

Extreme temperatures, extreme overhead
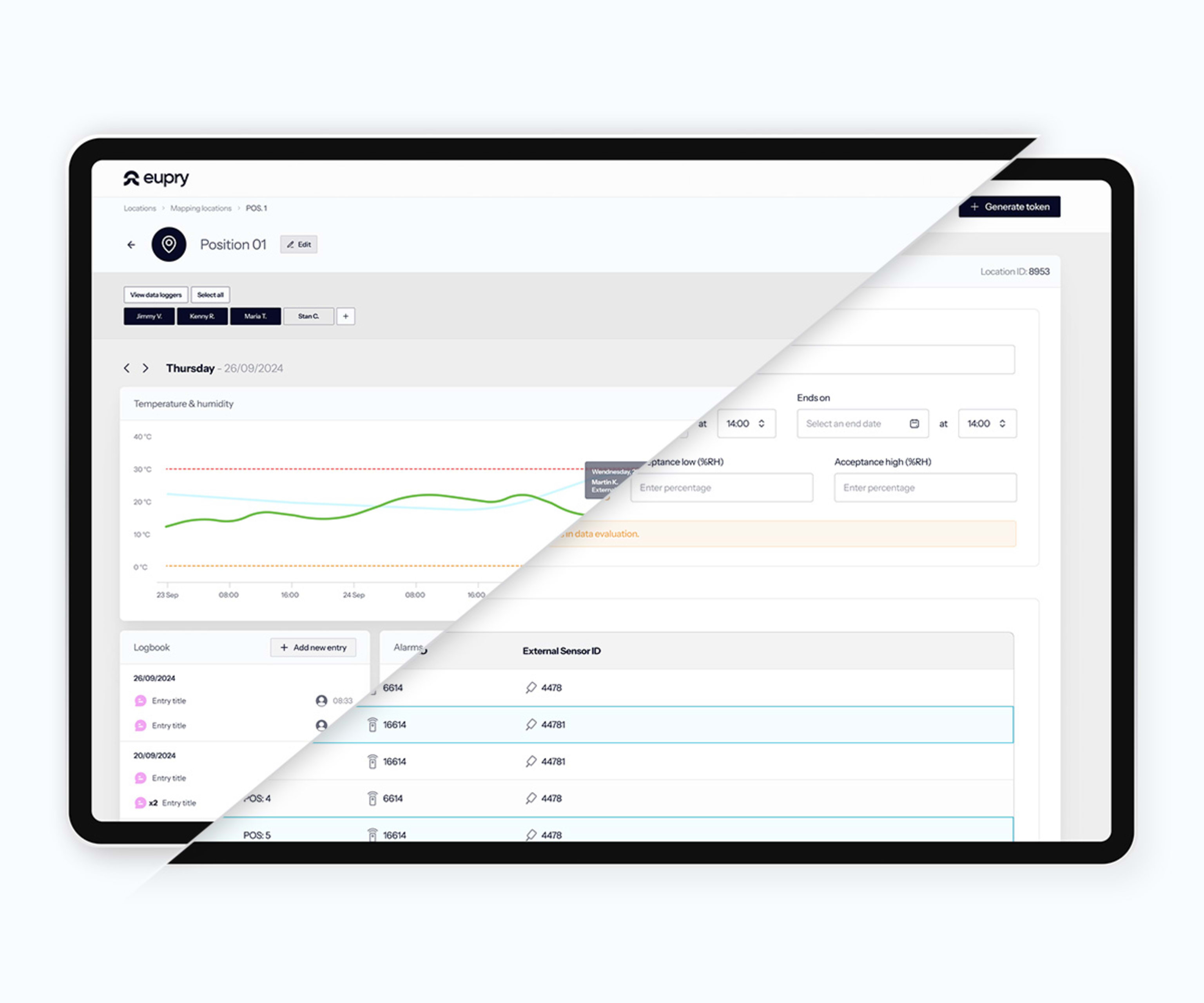
Most organizations rely on manual checks, fragmented systems, and multiple vendors for ULT and cryo compliance. Legacy systems lack redundancy, and a single overnight failure can destroy thousands of dollars in product. Extreme temperatures add complexity: thick sensor cables cause frosting and door seal failures. Traditional calibration requires full defrost, product relocation, and days of hazardous work.

One platform for extreme temperatures
Eupry monitors ULTs and cryos continuously with SMS, email, and call alerts when temperatures drift. Generate audit-ready reports in 3 clicks, combining validation, monitoring, and calibration data with automatic traceability. ISO-accredited calibration happens on-the-wall in seconds while the unit stays in operation, with no defrosting required. Built-in redundancy ensures no single point of failure. Compliant with FDA, EMA, and more.


“Eupry was very professional and informative throughout the process, providing us with high-quality service (...) It's undoubtedly a well-put partnership.”
Joakim Sund
Quality Assurance Specialist

“Eupry was instrumental in helping us establish a GDP-compliant operation ready for the standards of pharmaceutical logistics. Their flexibility, expertise, and collaborative approach were exactly what we needed to navigate the requirements of the industry and develop a foundation that worked for us."

Michael O'Brien
President

“All of the loggers have proven their worth and saved precious samples on several occasions when freezers broke or were accidentally left open.“

Anja Pomowski
Senior Scientist Antibody Production at Antikor
Relevant products

Download our Product Catalog
Get an overview of how Eupry’s products can help with automatic monitoring and mapping of temperatures, humidity, CO2 and diff. pressure.
Want to know more?
Discover how Eupry's automated compliance (monitoring, calibration, and validation) solutions work today.