
Featured article
The flying warehouse case
Continuous aircraft mapping and monitoring is changing the pharmaceutical cold chain, turning aircraft into validated environments, saving millions, and minimizing the risk to products.
Learn the basics of the "flying warehouse" concept.

Articles

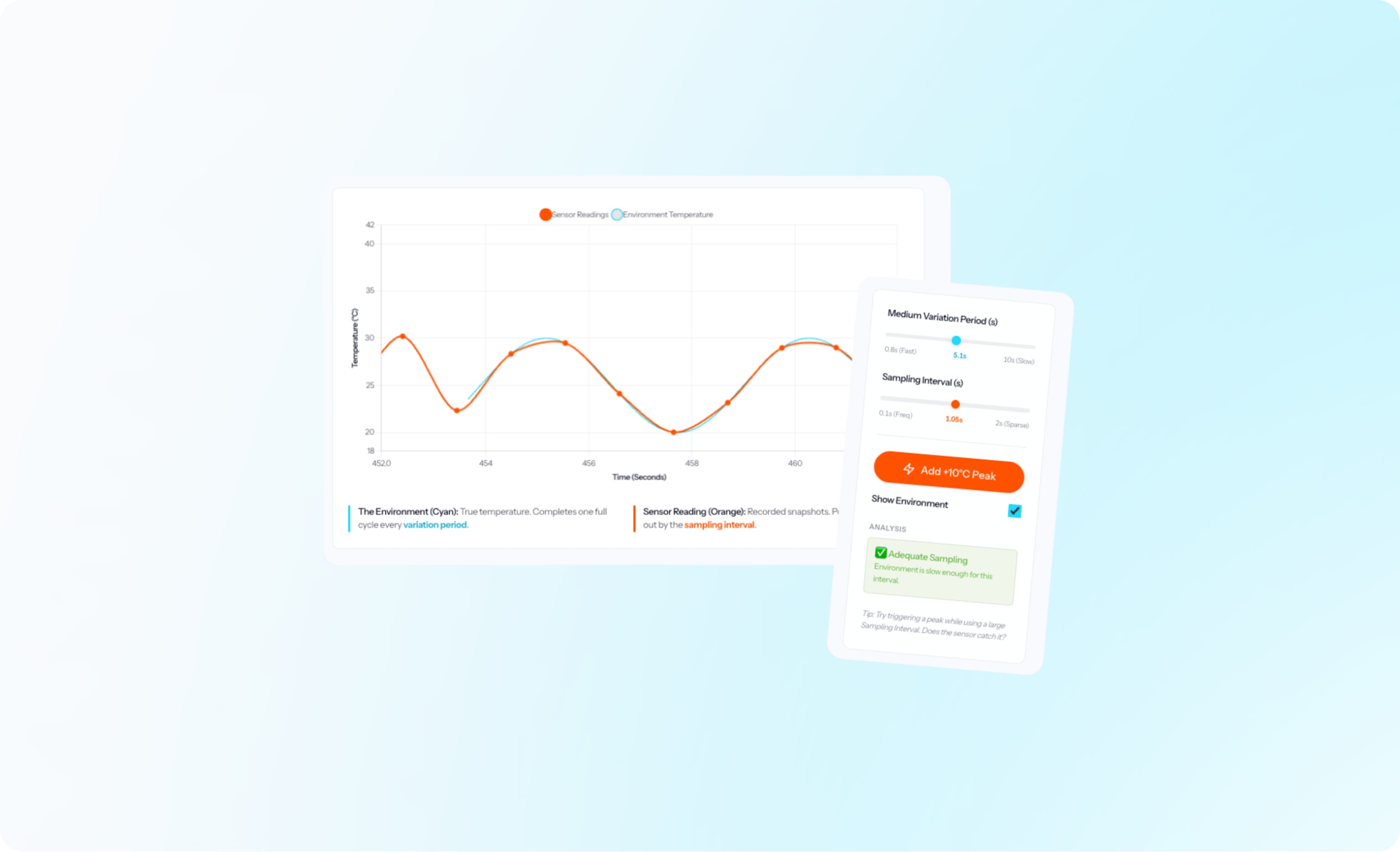
How to choose a sampling interval for mapping

What is dual compliance in GxP?

Guidelines for humidity monitoring in pharmaceutical environments
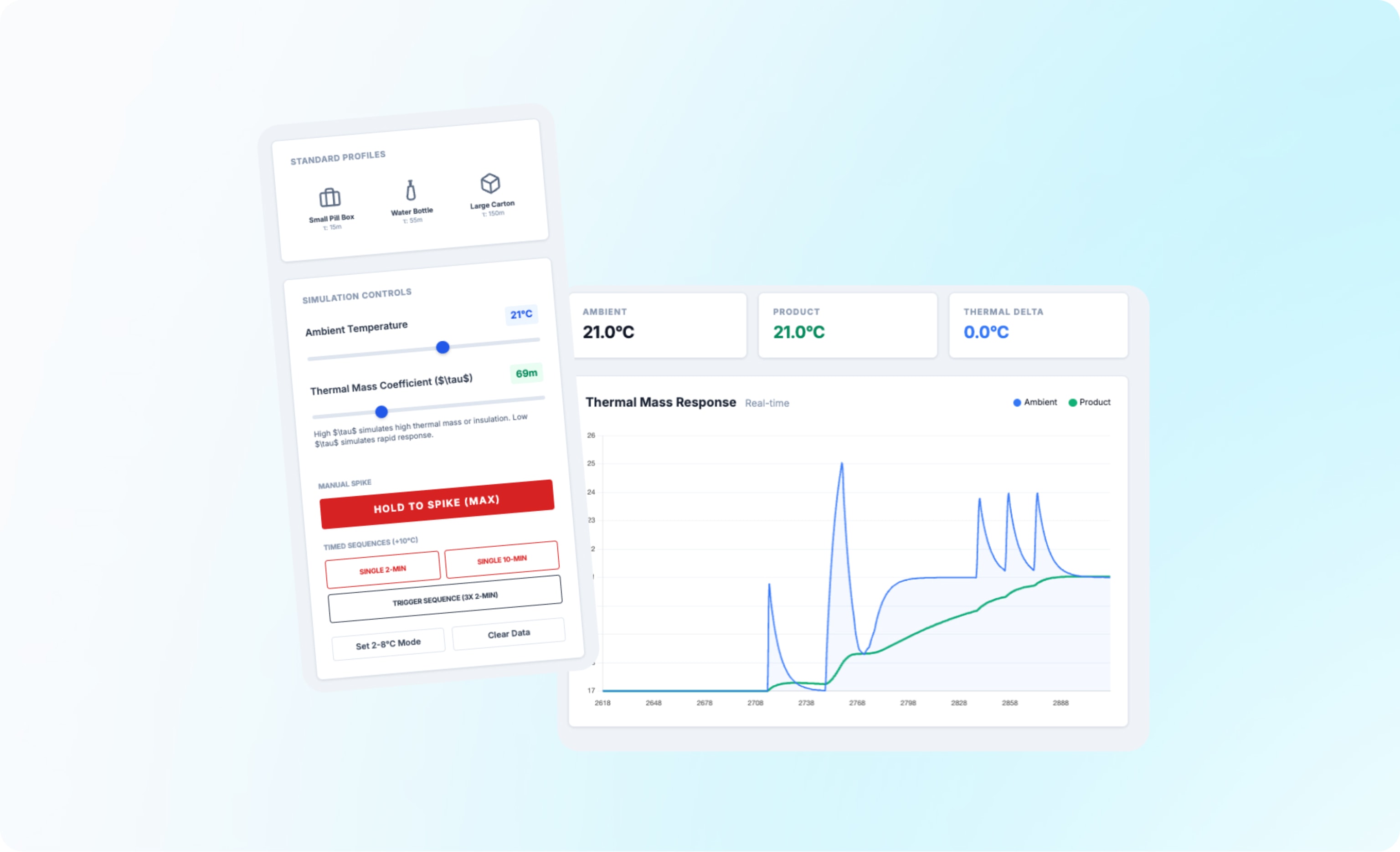
Thermal mass, thermal lag, and product integrity
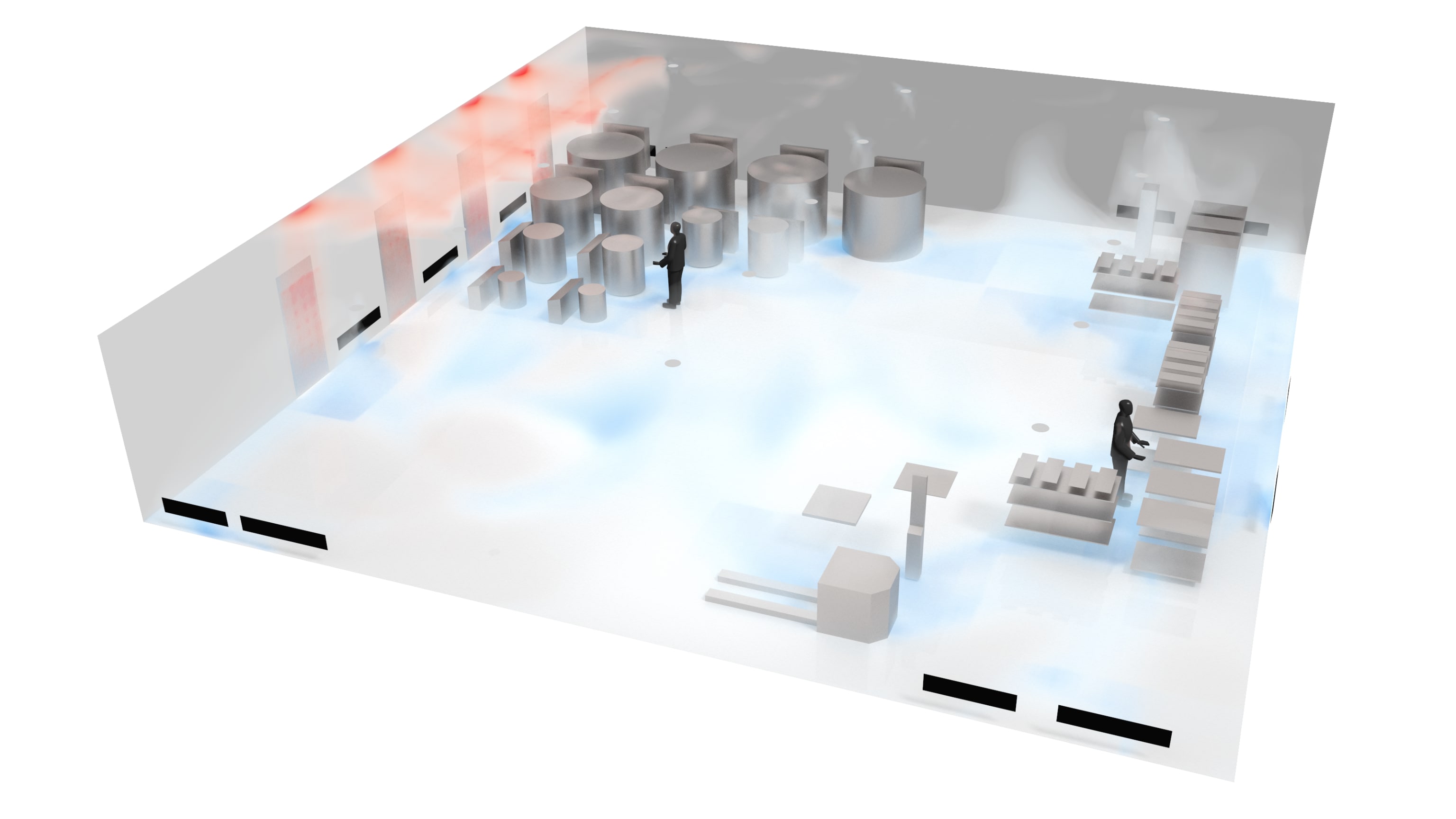
Differential pressure monitoring in pharmaceutical cleanrooms
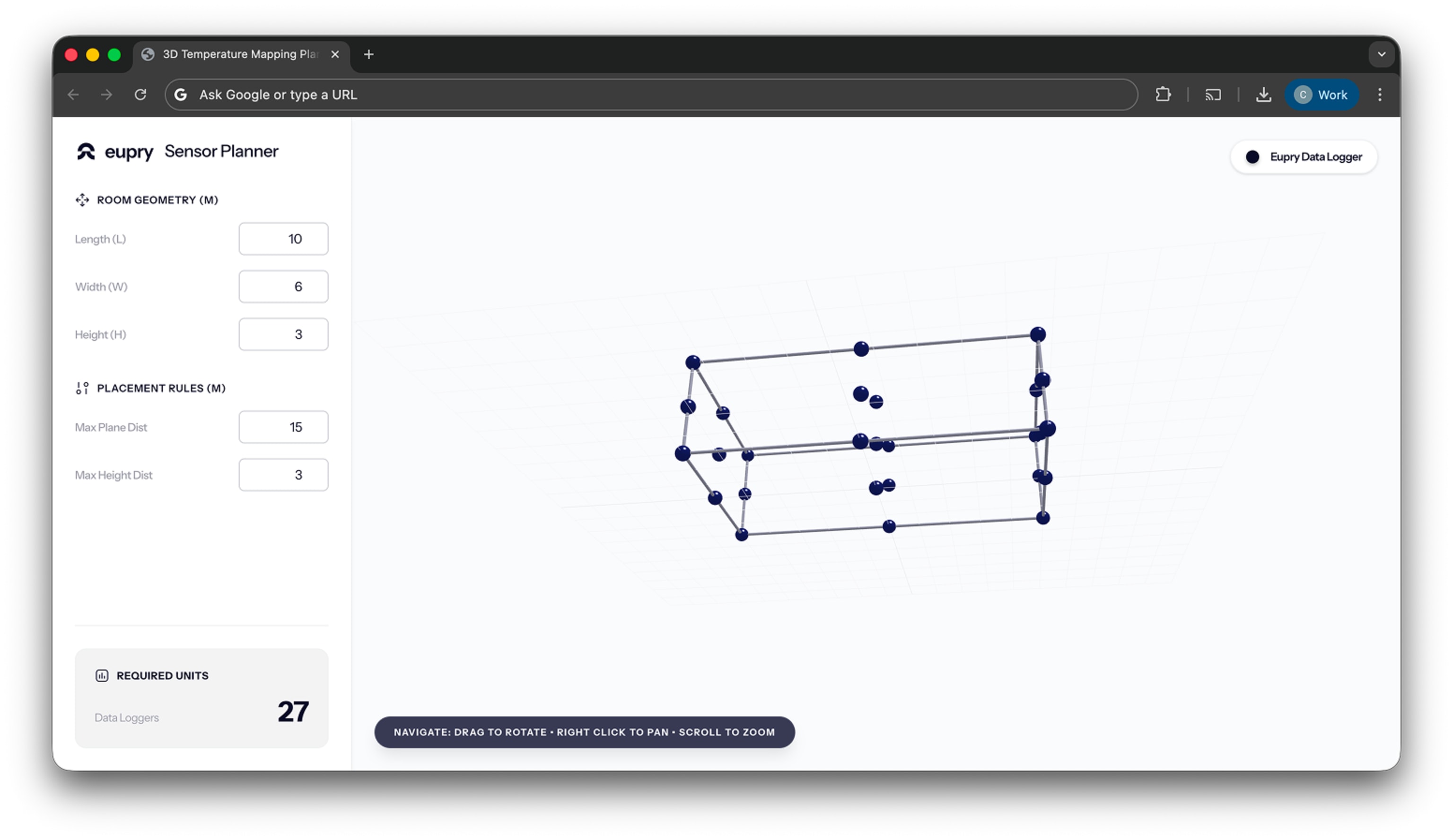
A guide (+ a tool) to 3D data logger placement in GxP mapping

The flying warehouse concept
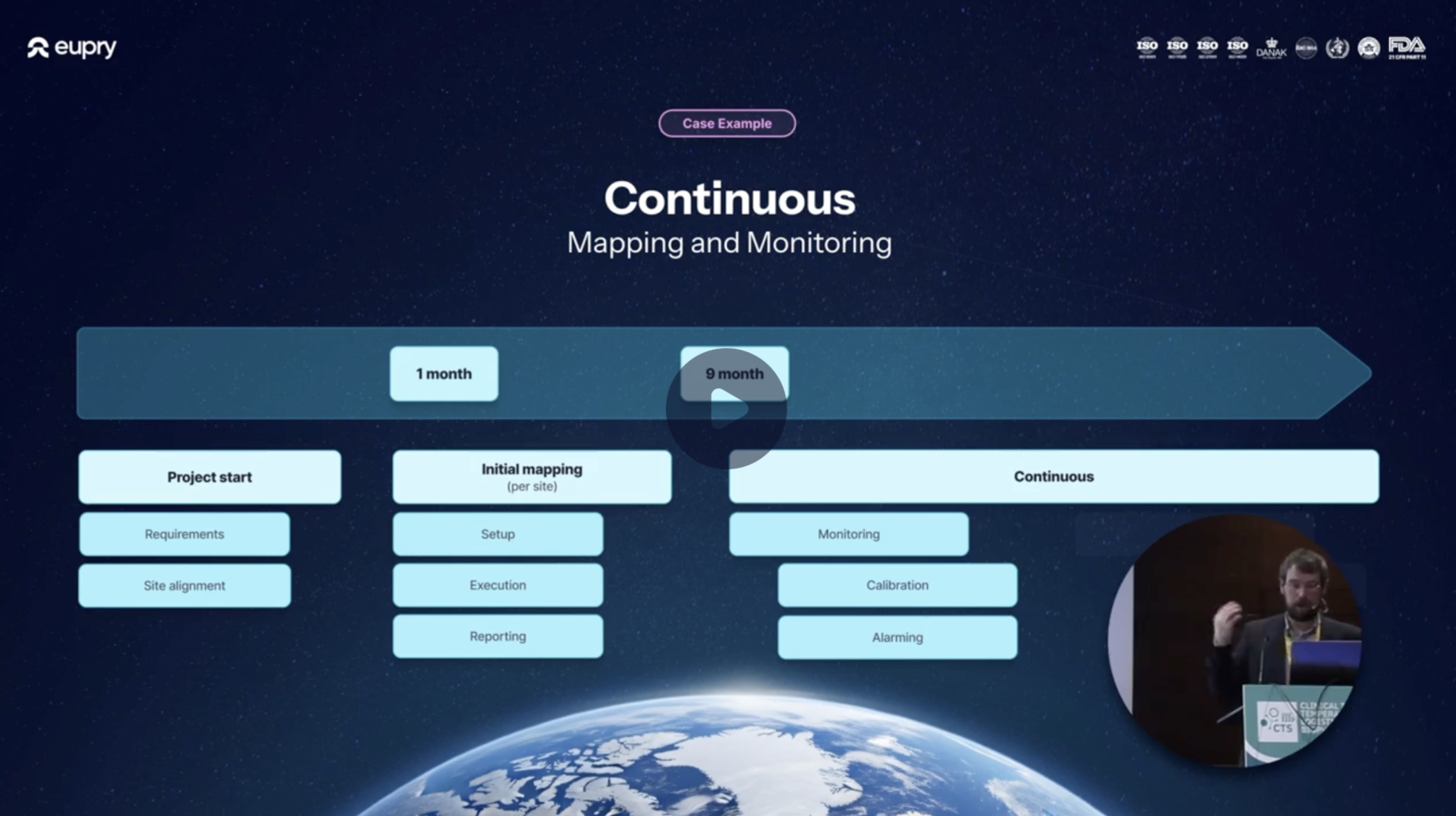
Continuous mapping (and monitoring) in practice

What QMSR means for labs using incubators

What is ISPE Validation 4.0?

Lyophilizer validation guide
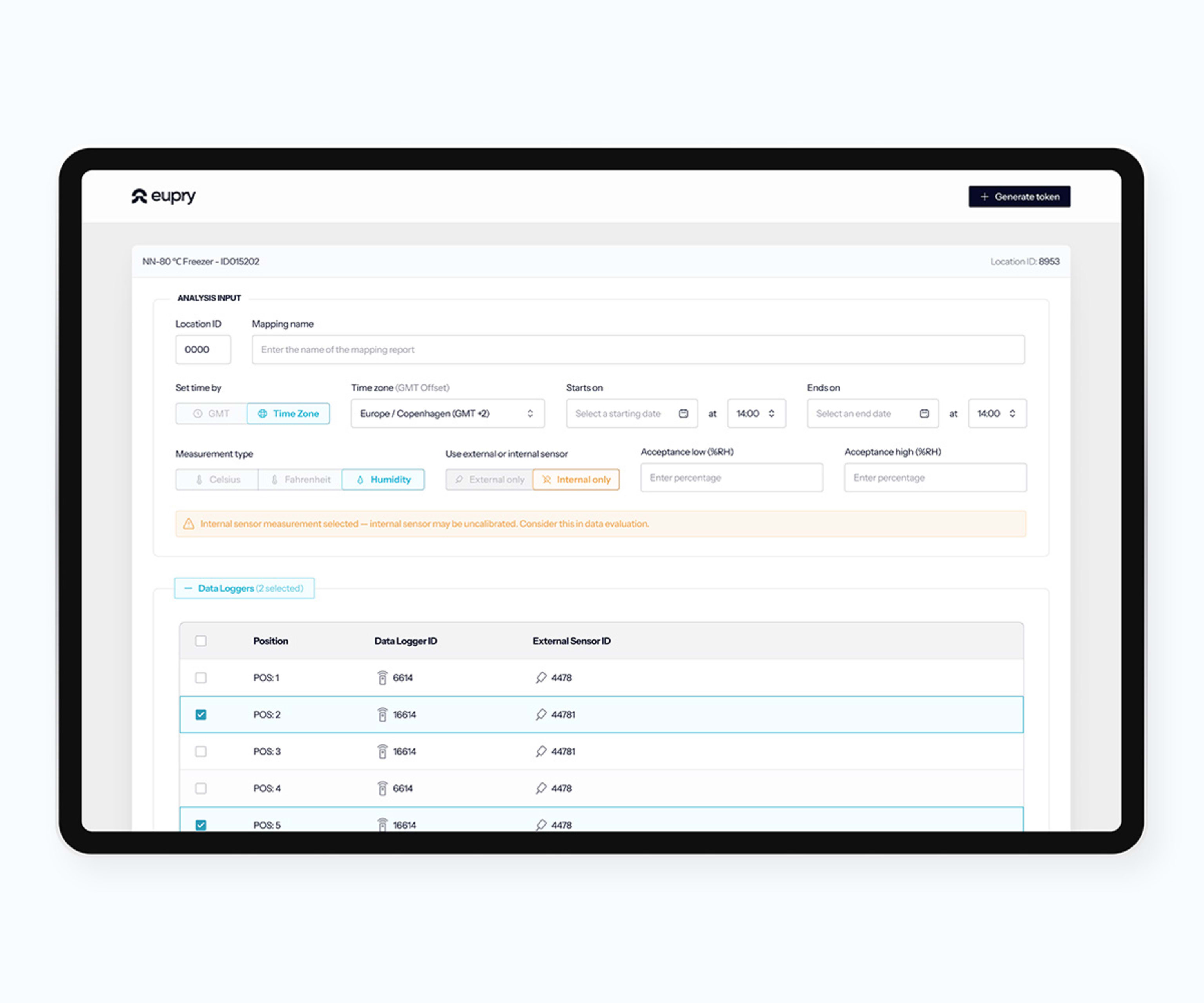
How to read temperature mapping data

Digital Calibration Certificates (DCC)

How to launch facilities before the second seasonal mapping

What to do when temperature mapping fails partially

What is pharmaceutical shipment monitoring?

The future of cold chain monitoring?

Pharmaceutical cold chain monitoring

Autoclave validation guide

6 steps to standardize thermal compliance
More resources
Guides and checklist

Guides and checklist
Checklists, guides, templates, and more to help enhance your temperature compliance processes – from monitoring to mapping calibration.
Webinars

Webinars
Watch live and on-demand webinars about everything from hands-on mapping guidelines to automating your compliance practices.
GxP glossary

GxP glossary
Get a quick overview of the key terminology of temperature compliance and GxP – what it means, when it applies, and why it matters.