What is dual compliance in GxP?
Dual compliance is a cold chain monitoring approach that combines continuous environmental monitoring of facilities and fleet with shipment-level tracking – giving pharmaceutical logistics teams the ability to protect products in transit and prove compliance for every shipment, without putting a device on every package. The goal is end-to-end visibility across the full cold chain, from raw material to patient.

Table of contents
Why is dual compliance relevant in GxP?
The pharmaceutical industry loses an estimated $35 billion annually to cold chain failures, according to IQVIA data reported by Forbes. That is medicine that never reaches the patient. Most of it preventable.
The losses persist because the traditional monitoring model was designed to document what happened – not to prevent it.
What are the problems with traditional cold chain monitoring?
In a traditional setup, a data logger or smart label is attached to each shipment. It records temperature throughout the journey and delivers the data after arrival – or, in the case of connected devices, in real time.
This gives you one dataset: What happened to the product.
That data is useful for compliance documentation. But it has three limitations that get worse as operations scale.
It is reactive, not preventive. Most devices deliver data after delivery. By the time you see an excursion, the product is already compromised. Even when the data is live, alerts often go to the shipper – not the carrier personnel who could actually intervene.
It does not explain the cause. When an excursion shows up on a shipment logger, you cannot tell whether the warehouse failed, the aircraft failed, or the product was mishandled. You see that something went wrong, but not where or why. Without that distinction, you are guessing at what to fix.
It does not scale. Every shipment needs its own device at $20 – 200 each. More shipments mean more devices, more cost, and more complexity. Compliance costs grow linearly with volume.
How does dual compliance work?
Dual compliance adds a second, independent data layer: continuous environmental monitoring of the transit environments the product passes through.
The two layers
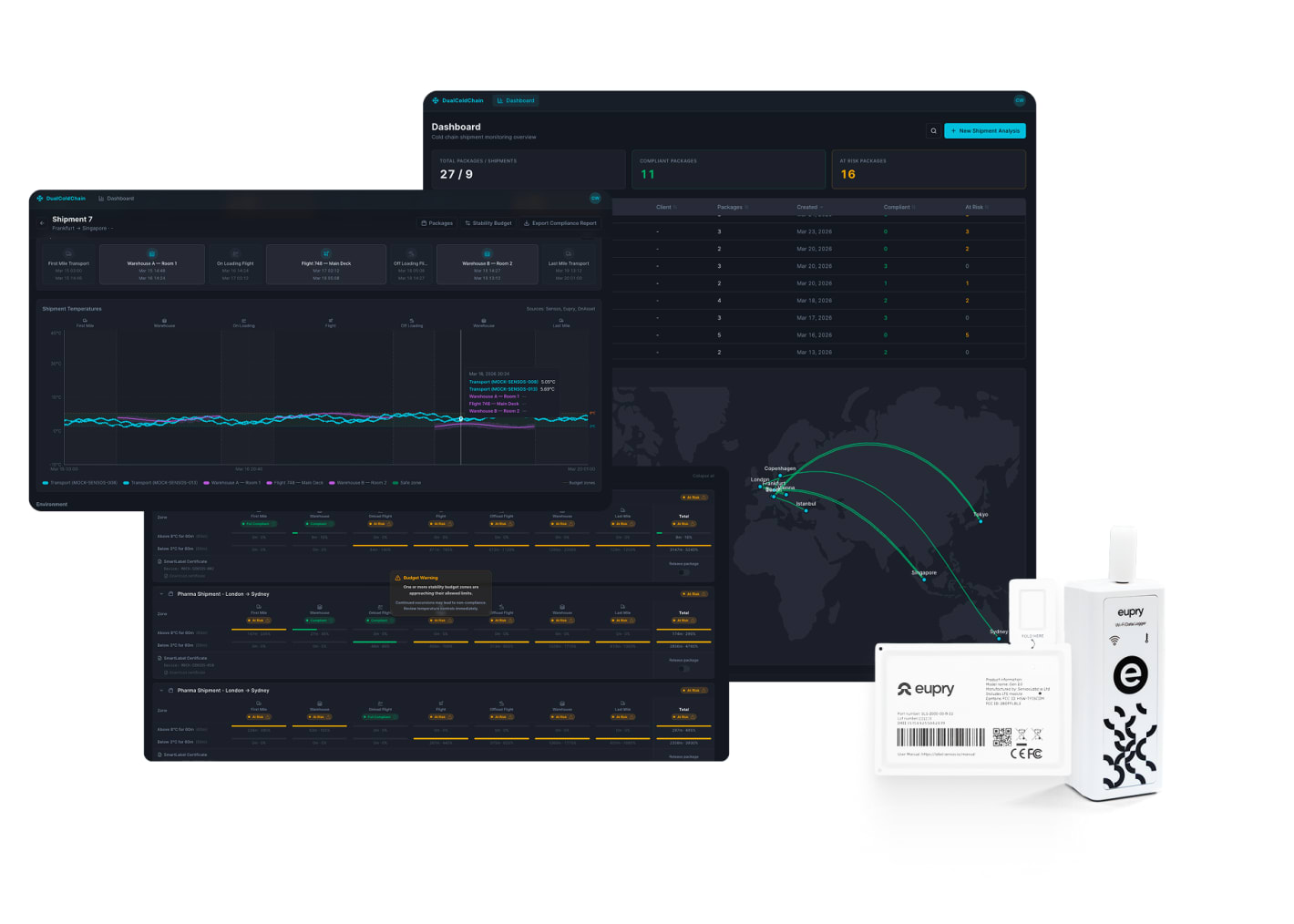
Layer one – environmental monitoring: Wireless data loggers installed permanently in warehouses, aircraft cargo holds, and trucks measure conditions continuously – typically every three minutes. This creates a validated environmental record for every product that passes through, whether or not it has its own tracking device.
A critical part of this layer is the aircraft. Warehouses have traditionally been mapped and monitored, but aircraft cargo holds have been a compliance blind spot – forcing operators to overcompensate with expensive active ULDs or accept data gaps. Continuous mapping and monitoring for aircraft changes that by treating the plane as a validated environment, with FAA-approved sensors installed permanently in the cargo hold. Data is available within minutes of landing, not days after delivery.
Layer two – shipment tracking: Smart Labels attached to high-risk or high-value shipments track temperature, humidity, GPS, shock, and tamper events in real time. These go on the shipments that actually need individual monitoring – not on every package by default.
Dual compliance vs. traditional shipment monitoring: What changes?
When both layers are in place, the compliance model shifts in three ways.
You can act before products are affected. Environmental data is live. When conditions in a warehouse or aircraft trend toward limits, carrier personnel are alerted and can adjust handling before the product's stability budget is exceeded. You prevent failures instead of documenting them.
You can prove compliance without monitoring every shipment. The environmental data provides a compliance baseline for everything passing through your facilities and fleet. Individual tracking is reserved for shipments where risk demands it – high-value biologics, new routes, or regulatory-specific requirements. Both data layers merge into one automatically generated report per shipment, with environmental and shipment-level data correlated across every handoff point. No manual data stitching, no spreadsheets, no separate systems.
You can pinpoint the actual cause of excursions. When you have environmental data from the warehouse, the aircraft, and the truck alongside the shipment data from the Smart Label, you can see exactly which phase caused the problem. Not "somewhere between A and B" – but in which facility, during which window. Over time, patterns emerge: this route has issues in summer, and that handoff point is consistently problematic. Root cause analysis becomes the basis for preventing the next excursion, not just investigating the last one.
Also read: From shipment to facility control: the future of cold chain monitoring
- | Traditional shipment monitoring | Dual compliance |
Data source | One: the device on the shipment | Two: environmental sensors in facilities and fleet + device on the shipment |
When you get data | After delivery (USB loggers) or live, but to the shipper (connected loggers) | Live, to the personnel handling the product |
What you can do | Document what happened after it is too late to intervene | Intervene before products are affected |
Root cause analysis | Limited – you see the excursion, not the cause | Clear – you can compare facility data to shipment data and pinpoint where the issue originated |
Compliance coverage | Only shipments with a device attached | Every product passing through a monitored facility, plus individual tracking where needed |
Cost model | Variable – scales linearly with shipment volume | Fixed environmental monitoring + selective shipment tracking |
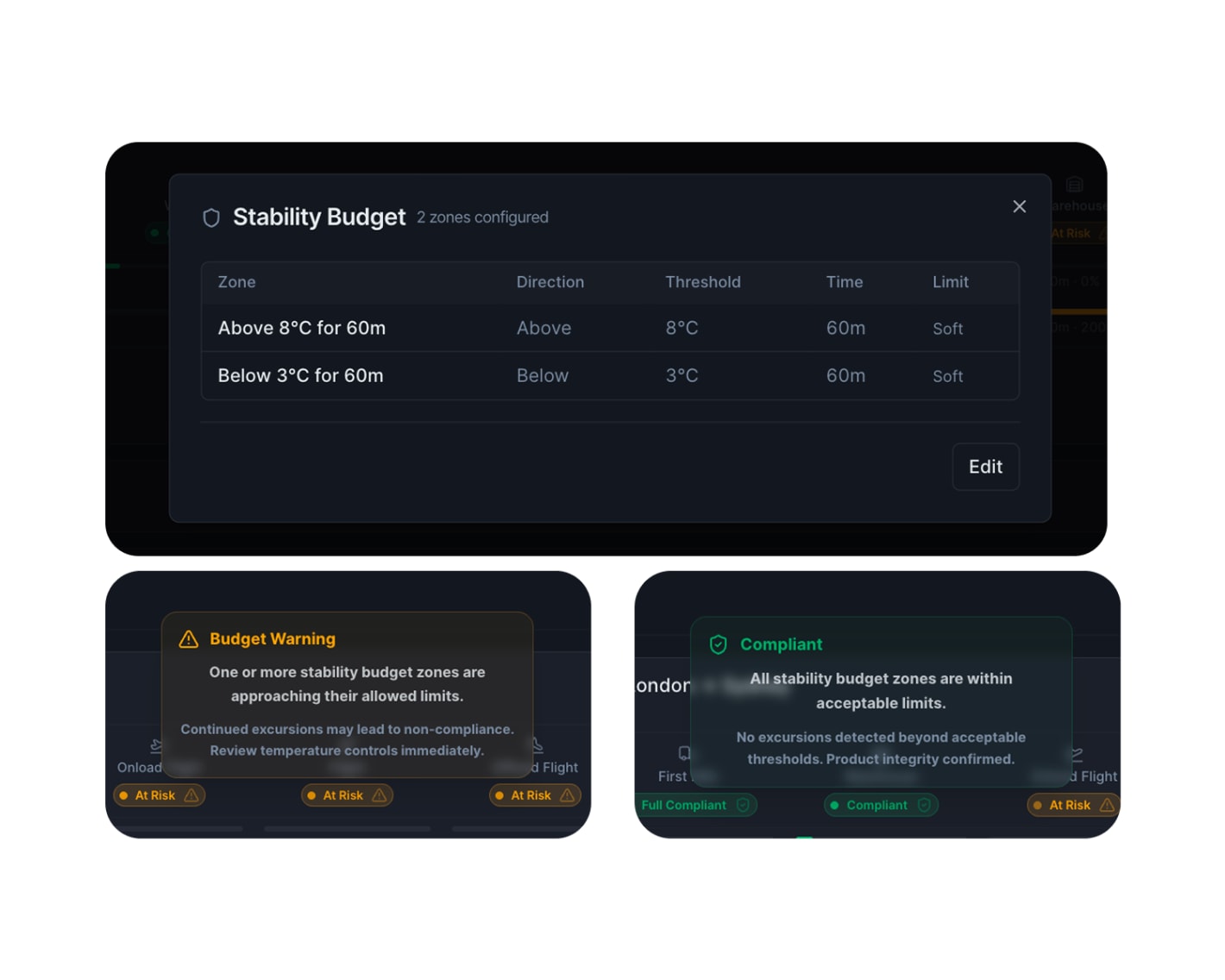
What is a stability budget?
A stability budget defines how long a product can spend outside its required temperature range across the entire journey – not just within a single phase.
For example, a product might have a budget of 60 minutes outside +2 °C to +8 °C (36 °F to 46 °F). If the product spent five minutes outside range in a warehouse and 35 minutes outside the range on an aircraft, the cumulative total is 40 out of 60 minutes. The shipment is compliant.
The critical point: each individual phase might look fine on its own. But the cumulative total across all phases can exceed the budget without any single phase triggering an alarm. Without a system that tracks this automatically across the full journey, that shipment could be released when it should not be.
In a dual compliance setup, stability budgets are tracked in real time across every leg – from raw material to patient. The teams handling the shipment see how much margin remains and can act before the budget runs out.
What is the difference between dual verification and dual compliance?
Dual verification and dual compliance are related but distinct concepts.
Dual verification is the transition methodology. It means running both environmental monitoring and shipment-level monitoring in parallel during an initial validation period – comparing the two datasets to build statistical evidence that facility-level data provides equivalent assurance to product-level monitoring.
Dual compliance is the ongoing operating model. Once dual verification has established confidence in the environmental data, the organization operates with continuous facility monitoring as the baseline and individual shipment tracking reserved for high-risk scenarios.
In practice, most organizations begin with dual verification and transition into dual compliance as their correlation data grows.

When do you still need individual shipment monitoring?
Dual compliance does not eliminate shipment-level tracking. It makes it strategic instead of universal.
Individual Smart Labels are still appropriate for high-value biologics or cell and gene therapies, shipments on new or unvalidated routes, products with specific regulatory requirements for shipment-level data, and scenarios where the transit environment is not yet continuously monitored.
The difference is that you choose which shipments get individual tracking based on risk, not because you have no other option.

What regulatory frameworks support dual compliance?
Dual compliance aligns with existing regulatory guidance on continuous process verification.
WHO TRS 961 (2021) endorses continuous verification as superior to periodic point-in-time snapshots for temperature-controlled environments.
FDA Process Validation Guidance (Stage 3) describes ongoing process verification as an approach to maintaining a validated state through continuous monitoring – the same principle underlying continuous environmental monitoring across facilities and fleet.
EU GDP guidelines require documented evidence that storage and transport conditions are maintained within validated ranges. Dual compliance provides this evidence through continuous environmental data rather than through individual shipment logs alone.
No regulatory guidance currently mandates dual compliance specifically. But the framework supports it – particularly when organizations can demonstrate through dual verification that their environmental monitoring provides equivalent or superior assurance to universal shipment monitoring.
Also read: What is GxP in pharma?
Frequently asked questions about dual compliance
Eupry's Dual Compliance Platform
Combine facility and shipment data
Know instantly when issues arise anywhere in the cold chain with the Dual Compliance Platform.
- Track stability budgets live across the full journey
- Act before products are compromised
- Prove GDP compliance (without tracking every shipment)
- Reduce per-shipment compliance cost