How to meet GMP temperature requirements
Annex 15 and Annex 11

Adam Hartmann-Kruckow
Learn what GMP requires for temperature mapping, validation, and monitoring to stay compliant. Get practical guidance on Annex 15 qualification, Annex 11 data integrity, and audit-ready documentation.
Get an instant overview of the key areas regulators typically examine during GMP inspections.

The short version: What are the thermal compliance requirements for GMP?
Good Manufacturing Practice (GMP) requires you to qualify temperature-controlled storage areas before use, monitor conditions continuously with validated systems, and maintain complete audit trails that prove data integrity. EU GMP Annex 15 governs physical qualification through IQ/OQ/PQ and temperature mapping studies. Annex 11 establishes electronic records requirements, including ALCOA+ data integrity principles. Together, these regulations ensure you store pharmaceuticals under validated conditions that protect product quality and patient safety.
Also read: Key regulations of temperature compliance
Note
This guide explains what GMP requires for temperature compliance and how to meet those requirements. It is meant for educational purposes only. It does not replace internal SOPs or risk assessments. Always confirm decisions with your quality unit and manufacturer. You will learn how to qualify storage areas (Annex 15), validate monitoring systems (Annex 11), maintain audit trails, and prepare for regulatory inspections. Whether you are setting up your first validated facility or managing compliance across multiple sites, this guide covers the practical steps you need for GMP compliance.
What is in this guide
- How to use this guide
- What is GMP and why does it matter for temperature control?
- 5 GMP principles for temperature control
- Why GMP temperature compliance is important
- What does GMP Annex 15 require for validation and mapping?
- What does GMP Annex 11 require for monitoring systems?
- How do Annex 15 and Annex 11 work together?
- GMP temperature requirements by regulatory authority
- Common GMP challenges and how to address them
- How Eupry supports GMP compliance
- Frequently asked questions about GMP

How to use this guide
-
If you are new to GMP temperature compliance: Start with "What is GMP and why does it matter" to understand the basics, then read through each section in order.
-
If you need to validate a specific system: Jump to "What does Annex 15 require" for qualification requirements, or "What does Annex 11 require for monitoring systems".
-
If you are preparing for an audit: Read "How do I demonstrate GMP temperature compliance during an audit" in the FAQ section and "Common challenges and how to address them."
-
If you are comparing requirements: Check "GMP temperature requirements by regulatory authority" to see how FDA, EMA, and WHO expectations differ.
-
If you want practical tools: Download the compliance checklist and look for the actionable checklists throughout each section.
What is GMP, and why does it matter for temperature control?
Good Manufacturing Practice (GMP) is a quality system ensuring medicinal products are consistently manufactured and controlled according to quality standards. GMP applies to the entire pharmaceutical lifecycle, from raw materials through production, storage, and distribution to end users.
For temperature-sensitive products, GMP compliance means demonstrating through qualification, validation, and ongoing monitoring that storage areas and equipment maintain required conditions. Failure to meet GMP temperature requirements can lead to product degradation, regulatory action, and risks to patient safety.
GMP requirements are enforced globally by authorities including the European Medicines Agency (EMA), FDA, WHO, and national regulators. In Europe, GMP guidance is published as EudraLex Volume 4, which includes critical annexes for temperature compliance.
Also see: What are the GxPs in pharma: The key components of GxP compliance
5 GMP principles for temperature control
- Qualification before use: You cannot store products in any temperature-controlled area until it is been qualified and shown to maintain specified conditions.
- Validation of systems: Your monitoring equipment and software must be validated to prove they capture accurate, reliable temperature data.
- Data integrity: All temperature records must meet strict standards (ALCOA+) to ensure they are trustworthy, complete, and tamper-proof.
- Continuous monitoring: Real-time monitoring with calibrated devices detects deviations immediately, enabling corrective action before products are affected.
- Documentation: Complete records of qualification studies, monitoring data, calibration certificates, and deviation investigations must be maintained and readily available for audits.
Also see: Requirements and guidelines for Good Distribution Practice (GDP)


Download an audit checklist for GMP
What do auditors look for in your temperature monitoring, mapping, and calibration processes?
Get an easy overview of the key areas regulators typically examine during GMP inspections.
Why is GMP temperature compliance important?
Temperature excursions during storage or transport can degrade active pharmaceutical ingredients, reduce product efficacy, or render medicines unsafe. GMP temperature requirements exist to prevent these risks through systematic controls.
What happens if you are not compliant
Regulatory consequences:
- Warning letters and observations from FDA, EMA, or other authorities
- Facility shutdowns or manufacturing holds
- Loss of manufacturing authorization
- Product recalls and disposal costs
Business impact:
- Delayed facility launches (cannot start operations until validated)
- Lost revenue from unsaleable product
- Customer contract losses (pharma clients require GMP compliance)
- Damage to reputation and market position
Patient safety:
- Ineffective medications reaching patients
- Potential adverse events from degraded products
- Erosion of trust in the pharmaceutical supply chain
The cost of non-compliance vs. compliance
A single temperature excursion affecting high-value biologics can cost hundreds of thousands in product loss. Regulatory observations require expensive remediation, consultant fees, and validation backlogs. In contrast, systematic GMP compliance through qualified systems and validated monitoring costs significantly less and prevents these risks entirely.
What does GMP Annex 15 require for temperature validation?
EU GMP Annex 15 (Qualification and Validation) establishes the framework for validating temperature-controlled equipment, facilities, and processes in pharmaceutical manufacturing. Published in 2015 and effective since October 2015, Annex 15 emphasizes a lifecycle approach and risk-based qualification.
Annex 15 mandates the qualification of all equipment and facilities before use and requires ongoing verification throughout their operational life. For temperature-controlled areas, this means systematic documentation that storage conditions remain within validated limits.
The qualification lifecycle covers
User Requirement Specification (URS): Define what the system must achieve, including temperature ranges, acceptable variation, and alarm thresholds before qualification begins. This is your baseline for all testing.
Design Qualification (DQ): Verify that the design of storage areas, refrigeration systems, and monitoring equipment meets the URS and GMP requirements.
Installation Qualification (IQ): Document that equipment is installed correctly, calibration certificates are in place, and utilities (power, refrigerant, sensors) are properly connected.
Operational Qualification (OQ): Test that equipment operates within specified parameters across its full operating range, including alarm functionality, temperature distribution, and recovery from door openings or power interruptions.
Performance Qualification (PQ): Demonstrate under actual operating conditions that the system consistently maintains required temperatures, typically over a minimum period defined by risk assessment (often 24-72 hours for refrigerators, 7-14 days for warehouses).
Also see: Guide to IQ, OQ, PQ in pharmaceuticals


Download a free IQ/OQ/PQ protocol pack for pharma
Get instant access to an all-in-one protocol pack: From URS checklists to IQ, OQ, and PQ protocol templates. All you need to plan the qualification of temperature-controlled equipment and environments in pharmaceuticals, biotech, and logistics, aligned with WHO, FDA, GMP/GDP/GxP guidelines.
GMP Annex 15
What does Annex 15 require for temperature mapping?
Temperature mapping is the critical study that demonstrates uniform temperature distribution within a controlled space. Annex 15 requires mapping to identify hot and cold spots, confirm that all locations remain within limits, and determine optimal placement for monitoring sensors.
Mapping requirements:
- Before initial use: Map every temperature-controlled storage area before storing any product in it.
- Risk-based sensor placement: Cover worst-case locations including doors, air inlets, corners, top and bottom shelves. WHO guidelines suggest approximately 1 sensor per 5-10 meters in small controlled spaces, with closer spacing (1 per 2-5 meters) for areas with greater variability.
- Documented protocol: Create a protocol with predefined acceptance criteria and rationale before starting the study. The protocol should define temperature ranges, maximum allowable variation, study duration, and sensor locations.
- Sufficient duration: Capture representative conditions and any cyclical variations. Refrigerators and freezers typically require 24-72 hours minimum. Warehouses need 7-14 days to capture loading cycles, door traffic, and environmental variations.
- Requalification triggers: Remap after significant changes to layout, HVAC systems, loading patterns, product types, or following repeated temperature excursions.

Annex 15 compliance checklist
- User Requirement Specification (URS) defining temperature ranges and alarm thresholds
- Design Qualification (DQ) verifying the system design meets requirements
- Installation Qualification (IQ) documenting correct installation and calibration certificates
- Operational Qualification (OQ) testing alarm functionality and temperature distribution
- Performance Qualification (PQ) demonstrating consistent performance
- Temperature mapping study identifying hot and cold spots
- Risk assessment determining requalification frequency
- Documented procedures for handling deviations and requalification triggers
- Training records for personnel conducting qualification activities
- Validation report summarizing all qualification activities and accepting the system for use
When do you need to requalify temperature systems under Annex 15?
Annex 15 introduced a requalification chapter recognizing that equipment must be periodically evaluated to confirm it remains in a validated state.
For temperature systems, requalification is triggered by:
- Changes to refrigeration systems, HVAC, or control software
- Modifications to room layout, shelving, or storage capacity
- Out-of-specification results or repeated temperature excursions
- Time-based schedules (typically annual or biennial reviews based on risk assessment)
- Seasonal variations affecting environmental conditions
An alternative to periodic requalification is continuous mapping that provides ongoing verification of temperature uniformity. This approach, recognized by ISPE Good Practice Guides and increasingly accepted by regulators, eliminates the need for disruptive remapping studies.
Also read: Continuous temperature mapping services for GMP
How does Annex 15 use risk-based qualification?
Annex 15 emphasizes that qualification is not a one-time activity but a continuous process throughout the equipment lifecycle. The 2015 revision explicitly states that retrospective validation is no longer an acceptable approach for new products or processes. Quality risk management (aligned with ICH Q9) should determine the scope and frequency of qualification activities based on:
- Criticality of the equipment to product quality
- Complexity of the system and potential failure modes
- Historical performance data and trend analysis
- Impact of environmental factors (seasonal variations, facility modifications)
This risk-based approach allows for proportionate validation strategies rather than rigid, prescriptive requirements. For well-understood, stable storage systems with robust continuous monitoring and continuous mapping, the validation burden can be significantly reduced compared to traditional periodic re-mapping.
GMP Annex 11
What does GMP Annex 11 require for temperature monitoring systems?
EU GMP Annex 11 (Computerised Systems) governs the use of digital systems in GMP-regulated activities. For monitoring, Annex 11 establishes requirements for electronic records, audit trails, system validation, and data integrity that complement the physical qualification requirements of Annex 15.
The current version of Annex 11 dates from 2011. A major revision (draft published in July 2025, expected final implementation mid-2026) introduces updated requirements for cloud systems, cybersecurity, AI/ML systems, and enhanced audit trail expectations.
Annex 11 applies whenever computerized systems are used to create, modify, maintain, or transmit GMP records. This includes temperature monitoring software
Annex 11 requirements for your monitoring system:
- System validation: The monitoring system must be validated before use, demonstrating it performs as intended and consistently generates accurate, complete temperature records. You need documentation showing the system does what it's supposed to do.
- Risk-based approach: Validation scope and controls should be proportionate to the system's GMP impact and the criticality of temperature data for product quality and patient safety.
- Audit trails: All changes to temperature data, user activities, system configurations, and calibration records must be captured in secure, time-stamped audit trails that cannot be disabled or edited by users.
- Access controls: Only authorized personnel should access the system, with unique user credentials (no shared passwords) and role-based permissions limiting who can view, modify, or approve records.
- Data integrity: Electronic records must meet ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) throughout the data lifecycle.
- Backup and disaster recovery: Temperature records must be backed up regularly, with tested procedures to restore data in case of system failures.
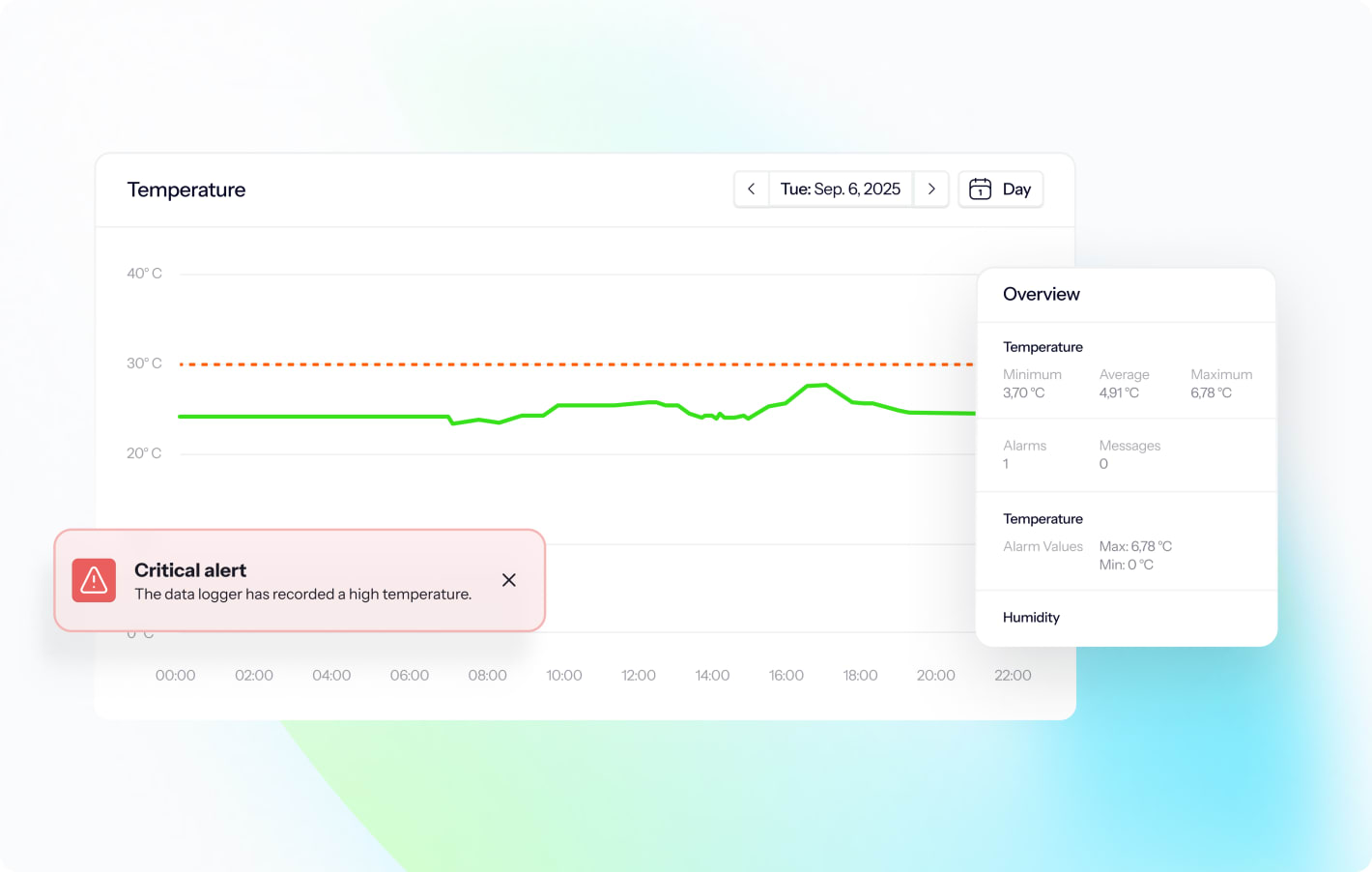
What is ALCOA+ and why does it matter for temperature data?
ALCOA+ defines what constitutes trustworthy data in GMP environments. For temperature monitoring, each principle translates to specific system capabilities and procedural controls:
- Attributable: Every temperature reading, alarm event, user action, and data change is linked to a specific person, device, or system process with date and time stamps. You must be able to answer "who recorded this data?"
- Legible: Temperature records remain readable throughout their retention period, in both electronic format and any required printouts, without degradation or format obsolescence.
- Contemporaneous: The monitoring system captures temperature data at the time of measurement (not transcribed later from paper notes) with accurate time stamps synchronized to a validated time source.
- Original: Temperature data is recorded directly by calibrated sensors to the monitoring system, not manually entered or transcribed from other sources unless specifically designed and validated for manual entry.
- Accurate: Data loggers are calibrated to traceable standards (ISO 17025), the monitoring system performs accuracy checks, and any data corrections are documented through formal change control with audit trail entries.
- Complete: All temperature measurements are retained, including out-of-specification readings, alarm events, system errors, and any deleted or modified data visible through audit trails. You cannot cherry-pick which data to keep.
- Consistent: Date and time formats, temperature units, measurement intervals, and data structures remain uniform across the system, with time stamps following a logical sequence.
- Enduring: Temperature records are stored on durable, validated media with regular backups, protected from accidental deletion or corruption, and remain accessible for the required retention period (typically 1 year beyond product expiry, often 5-10 years or longer for GMP facilities).
- Available: Authorized users can retrieve temperature records quickly for investigations, audits, or regulatory inspections, with search and reporting functions enabling efficient data access.
Annex 11 compliance checklist
Before using any computerized temperature monitoring system for GMP purposes, confirm:
- System validation documentation (IQ/OQ/PQ protocols) completed and approved
- Risk assessment documenting validation scope and critical functions
- Audit trails enabled and cannot be disabled by normal users
- All user accounts have unique credentials (no shared logins)
- Role-based permissions configured to limit access appropriately
- Electronic signatures configured (if used) with two-factor identification
- Backup procedures documented and tested successfully
- Data retention procedures meeting regulatory requirements (typically 5-10 years)
- Disaster recovery procedures documented and tested
- Calibration certificates for all sensors stored and linked in the system
- Change control procedures for system updates and configuration changes
- Training records for all users documented
- Periodic review schedule established (per draft Annex 11 requirements)
What changed in the July 2025 revision of Annex 11?
The July 2025 draft revision of Annex 11 significantly expands from 5 pages to 19 pages and introduces 17 sections (compared to 9 in the current version). The most substantial changes for temperature monitoring include:
Expanded audit trail requirements:
- Audit trails must be enabled at all times and cannot be disabled by normal system users
- Must capture not just changes and deletions, but also data creation (aligning closer to 21 CFR Part 11)
- Automated alerts should flag suspicious patterns (bulk deletions, after-hours modifications, repeated failed login attempts)
- Regular audit trail reviews must be documented, with defined frequency, scope, and responsibilities
- Reviews should be risk-based, focusing on critical data points and high-risk activities
Enhanced identity and access management:
- New dedicated section on access controls with multiple subsections
- Stronger requirements for unique user accounts and role-based permissions
- Better guidance on account lifecycle management
Mandatory periodic review:
- New requirement aligning computerized systems with equipment/facility periodic review expectations
- Must verify systems remain validated and fit for purpose at defined intervals
Cybersecurity requirements:
- Dedicated section on IT security (firewalls, patch management, virus protection)
- Disaster recovery expectations including RTO/RPO metrics
- Regular penetration testing required for critical systems
For monitoring, this means audit trails must capture not just data changes but also:
- User logins and logouts
- Changes to alarm thresholds or monitoring parameters
- Calibration certificate uploads or sensor replacements
- Data exports, report generation, and any access to historical records
- System configuration changes affecting how temperature data is collected or processed
Organizations should begin aligning monitoring systems with the draft expectations now, as retroactive changes to legacy systems can be complex and costly. The comment period closed on October 7, 2025, with final implementation expected in mid-2026.

How do 21 CFR Part 11 and Annex 11 relate?
If you operate in the US or supply to US companies, you also need to comply with FDA's 21 CFR Part 11 for electronic records and electronic signatures.
Part 11 has become a de facto global standard, often required even outside the US.
Key differences: Part 11 vs Annex 11
Geographic scope:
- FDA 21 CFR Part 11: US regulation, but globally influential
- EU GMP Annex 11: EU regulation, adopted by PIC/S member states
Risk-based approach:
- FDA 21 CFR Part 11: Less explicit; validation requirements apply broadly
- EU GMP Annex 11: Explicitly mandates risk-based validation scope
Audit trail requirements:
- FDA 21 CFR Part 11: Required for specific predicate rule activities
- EU GMP Annex 11: More comprehensive, covering all GMP-relevant changes
Legacy systems:
- FDA 21 CFR Part 11: Enforcement discretion for certain requirements
- EU GMP Annex 11: Full compliance expected; legacy systems must meet current standards
Lifecycle management:
- FDA 21 CFR Part 11: Focuses on static validation at implementation
- EU GMP Annex 11: Emphasizes continuous lifecycle management and periodic review
For more 21 CFR Part 11 guidance: Read our dedicated 21 CFR Part 11 guide
Most pharma companies design temperature monitoring systems to meet both Annex 11 and Part 11 simultaneously, ensuring global regulatory acceptability.


FDA 21 CFR Part 11-compliant temperature monitoring
Get a step-by-step checklist to make sure your temperature monitoring setup and system are FDA 21 CFR Part 11 compliant.
How do Annex 15 and Annex 11 work together?
Annex 15 and Annex 11 are complementary. Annex 15 defines what must be validated (temperature uniformity, equipment performance, alarm functionality), while Annex 11 governs how electronic systems generate and manage the data proving compliance.
A complete GMP temperature compliance program integrates both:
- Physical qualification (Annex 15): Temperature mapping studies prove storage areas maintain uniform conditions. Qualified equipment operates within specified parameters. Risk assessments determine requalification frequency.
- Digital validation (Annex 11): The monitoring software is validated to capture accurate data. Audit trails document all system activities. Access controls prevent unauthorized changes. Data integrity controls ensure ALCOA+ compliance.
- Continuous verification: Ongoing monitoring and mapping provides real-time evidence that qualified conditions persist. Automated alerts enable immediate corrective action. Trend analysis detects gradual drift before excursions occur.
Practical example of GMP compliance: Qualifying a new warehouse
Step 1 - Physical qualification (Annex 15):
- Create URS defining temperature range, maximum variation, alarm thresholds
- Perform IQ for installation of HVAC, refrigeration, and monitoring equipment
- Execute OQ testing alarm functionality, door recovery times, and distribution
- Conduct mapping study with 50-200 data loggers over 7-14 days
- Complete PQ under loaded conditions demonstrating consistent performance
- Document findings and accept warehouse for use
Step 2 - Digital validation (Annex 11):
- Validate the monitoring software used to collect and store data
- Configure audit trails to capture all data changes and user activities
- Set up user accounts with role-based permissions
- Establish backup procedures and test data recovery
- Create SOPs for monitoring, alarm response, and deviation handling
- Train users and document training completion
Step 3 - Ongoing compliance (both):
- Continuous monitoring and mapping at areas identified during initial mapping
- Regular calibration of monitoring sensors
- Audit trail reviews (monthly or quarterly based on risk)
- Investigation and documentation of any temperature excursions
- Requalification after any significant changes to the facility or equipment

GMP requirements by regulatory authority
GMP temperature requirements by regulatory authority
While GMP principles are globally harmonized, specific requirements vary by region and authority. Understanding these nuances ensures compliance across markets.
European Medicines Agency (EMA) – EudraLex Volume 4
EudraLex Volume 4 contains the EU GMP guidelines, including Annex 15 (Qualification and Validation) and Annex 11 (Computerised Systems). These are legally binding in EU member states and widely adopted by European Economic Area (EEA) countries and PIC/S member states globally.
Key EMA expectations for temperature compliance:
- Mapping required before use and after significant changes
- Risk-based approach to validation scope and frequency
- Lifecycle management with periodic review
- Comprehensive audit trails for electronic systems
- Emphasis on data integrity and ALCOA+ principles
EMA guidelines are often adopted verbatim by national authorities across Europe, creating regulatory consistency.
US Food and Drug Administration (FDA)
FDA GMP requirements are codified in 21 CFR Parts 210 and 211 (drug products) and Part 11 (electronic records). FDA emphasizes risk-based validation and process validation as a lifecycle activity (aligning with ICH Q8-Q11).
Key FDA expectations for temperature compliance:
- Equipment qualified and suitable for intended use (21 CFR 211.63)
- Temperature and humidity controls where needed to ensure product quality (21 CFR 211.46)
- Monitoring devices calibrated at appropriate intervals (21 CFR 211.160(b)(4))
- Complete documentation, including temperature records, retained per specified periods (21 CFR 211.180)
FDA inspection findings frequently cite inadequate temperature monitoring, missing calibration records, and insufficient investigation of excursions as GMP violations.
World Health Organization (WHO)
WHO Technical Report Series (TRS) provides guidance for countries without comprehensive national GMP frameworks. TRS 961 Annex 9 (2011) specifically addresses temperature mapping for storage areas.
Key WHO guidance includes:
- Temperature mapping required for all storage areas before use
- Risk-based sensor placement (minimum 1 sensor per 30 cubic meters for large warehouses, more in smaller areas with greater variability)
- Studies should cover seasonal variations where relevant
- Documented protocols, reports, and requalification schedules
WHO guidance is particularly influential in developing pharmaceutical markets and for WHO prequalification programs.
Also see: Guide: What are the WHO’s guidelines for temperature mapping?
International Council for Harmonisation (ICH)
ICH guidelines (Q8-Q11) provide harmonized standards adopted by FDA, EMA, and other regulators. While not specific to temperature, they establish principles for:
- Quality risk management (ICH Q9) to determine validation scope
- Pharmaceutical quality systems (ICH Q10) integrating validation into lifecycle management
- Development and manufacturing (ICH Q8, Q11) emphasizing process understanding over rigid protocols
These principles support modern validation approaches like continuous verification, which replace periodic re-mapping.
Challenges of GMP
Common GMP compliance challenges and how to address them
Even well-managed pharmaceutical operations face recurring challenges with GMP temperature compliance. Recognizing these pitfalls and implementing systematic solutions reduces risk and operational burden.
Challenge 1: Fragmented systems and multiple vendors
The problem: Many facilities use separate providers for data loggers, calibration services, mapping studies, and monitoring software. This fragmentation creates gaps in data traceability, complicates validation, and increases costs. Coordinating between vendors delays projects and creates compliance risks when data doesn't transfer cleanly between systems.
How to solve it: Consolidate temperature compliance functions (monitoring, mapping, calibration) with a single provider offering an integrated GxP-compliant platform. Unified systems simplify validation (one validation package instead of multiple), eliminate data silos, and provide a single audit trail across all temperature activities. You reduce vendor management overhead and get consistent support across all compliance needs.
Challenge 2: Manual processes and documentation gaps
The problem: Paper-based temperature logs, manual calibration tracking, and disconnected spreadsheets create data integrity risks, consume QA resources, and slow down investigations. Transcription errors occur when manually copying temperatures from loggers. Calibration certificates get lost in filing cabinets. Finding historical data for trend analysis takes hours or days.
How to solve it: Digital monitoring systems that automate data capture, calibration certificate storage, and audit-ready reporting. Systems meeting 21 CFR Part 11 and Annex 11 eliminate manual transcription, ensure ALCOA+ compliance, and generate reports in seconds rather than hours. Automated alerts notify you immediately of excursions instead of discovering problems hours later during manual log reviews.
Challenge 3: Lack of central visibility across sites
The problem: Global pharmaceutical operations struggle to maintain consistent standards and oversight when each facility uses different systems, processes, or vendors. Corporate QA can't see real-time compliance status across sites. Different facilities interpret GMP requirements differently. Audits require traveling to each site to review local records.
How to solve it: Cloud-based monitoring platforms that provide enterprise-wide visibility, standardized SOPs across sites, and centralized quality dashboards. This enables real-time monitoring of all facilities from one location, consistent alarming strategies, and streamlined audits. Corporate QA can review temperature data, calibration status, and deviation investigations for any site without travel.
Challenge 4: Costly and disruptive remapping cycles
The problem: Periodic temperature mapping studies require scheduling downtime, deploying data loggers, waiting days for results, and repeating the process every 1-3 years. This consumes validation resources and delays facility readiness. Equipment rental, consultant fees, and operational downtime add up to significant costs. Facilities can't operate during mapping studies.
How to solve it: Continuous mapping approaches that provide ongoing verification through permanently installed, calibrated monitoring sensors. Once the initial mapping establishes hot/cold spots, extended continuous monitoring at these risk locations eliminates re-mapping needs unless operations change significantly. You save time, reduce costs, and minimize risks.
See how the GMP-designed automated thermal compliance solutions work.

Challenge 5: Excursion investigations and deviation management
The problem: When [temperature excursions]](/temperature-monitoring/excursion-investigation/) occur, investigations require pulling data from multiple sources, interviewing staff, calculating Mean Kinetic Temperature (MKT), assessing product impact, and documenting corrective actions. Paper-based or manual processes make this slow and error-prone. Incomplete documentation leads to regulatory observations. Critical products may need to be destroyed unnecessarily due to lack of proper data.
How to solve it: Integrated monitoring platforms that automatically flag excursions, calculate MKT, generate investigation reports with complete audit trails, and link to CAPA systems. Automated alerts enable real-time response before products are affected. Complete data capture (who opened the door, how long it was open, ambient conditions) makes investigations faster and more thorough. Digital workflows guide investigators through root cause analysis.
Also see: Temperature excursion investigation guide
Challenge 6: Audit readiness and inspector requests
The problem: Auditors and inspectors expect immediate access to temperature records, calibration certificates, mapping reports, and deviation investigations. Scattered documentation across paper files, local servers, and personal drives creates delays and compliance risks. Finding a specific calibration certificate from three years ago takes hours. Demonstrating continuous monitoring coverage requires assembling data from multiple sources.
How to solve it: Centralized document management within the monitoring platform, where all temperature-related records (raw data, calibration certificates, validation protocols, investigation reports) are stored, indexed, and retrievable in seconds. Three-click audit reports demonstrate compliance instantly. Audit trails show complete chain of custody for all data. Calibration certificates are automatically linked to specific sensors and time periods.
Implementation roadmap: How to address these challenges
Immediate actions (0-30 days):
- Audit your current state: Map all vendors, systems, and processes
- Identify your biggest pain points from the list above
- Calculate current costs (vendor fees, staff time, operational downtime)
- Define requirements for an integrated solution
Short-term improvements (30-90 days):
- Select and validate a centralized monitoring platform
- Begin migrating critical facilities to the new system
- Standardize SOPs and procedures across sites
- Train users on new digital workflows
Long-term optimization (90+ days):
- Roll out continuous mapping to eliminate remapping cycles
- Implement enterprise-wide dashboards for corporate QA visibility
- Optimize calibration schedules using trend data
- Build historical data for predictive analytics and risk assessment
How Eupry supports GMP temperature compliance
Turning GMP requirements into practs
This guide covers what GMP Annex 15 and Annex 11 require for temperature compliance. Eupry's solutions put them into practice – providing the only GxP-compliant platform that unifies monitoring, validation, and calibration in one digital system.
We've helped 500+ pharmaceutical and logistics companies meet GMP and GDP requirements, from single-facility operations to global enterprises managing hundreds of sites.
This integrated approach simplifies GMP compliance, reduces costs by 25-30%, and ensures audit readiness across all facilities.
Meeting Annex 15 qualification requirements with Eupry
Eupry's ISO 17025-accredited mapping services execute IQ/OQ/PQ studies with pre-calibrated wireless data loggers and GxP-specialized software. After initial mapping identifies hot and cold spots, our sensors remain in place at these risk locations, providing continuous mapping and monitoring.
This continuous verification approach, recognized by ISPE, eliminates re-mapping and lowers costs by 20-30%.
Meeting Annex 11 and 21 CFR Part 11 data integrity requirements with Eupry
Eupry's platform is validated to meet both Annex 11 (including the July 2025 draft requirements) and comes with a specialized 21 CFR Part 11 modul. The system provides full audit trails capturing all user actions and data changes, role-based access controls with unique user credentials, and ALCOA+ compliant electronic records with automatic timestamping and tamper-proof storage.
Validation documentation (IQ/OQ/PQ protocols aligned with GAMP 5) is performed once centrally and deployed across all your sites, eliminating redundant validation work. Generate complete audit reports showing temperature data, deviatons, and calibration certificates in three clicks instead of spending days assembling documentation.
How Eupry's solutions solve common GMP compliance challenges
- Fragmented vendors: One platform for monitoring, mapping, calibration, and validation eliminates the compliance gaps and integration risks of managing multiple providers.
- Calibration burden: ISO 17025-accredited calibration is included at no extra cost. Pre-calibrated sensors arrive on schedule and snap into place in seconds without interrupting monitoring – reducing calibration time by up to 95%.
- Multi-site visibility: Enterprise dashboard provides real-time compliance oversight across all facilities, with centralized validation documentation and standardized processes.
- Validation backlog: Wireless deployment and digital protocols compress mapping timelines from weeks to days. Continuous monitoring enables immediate transition to operations without additional studies.


Automate GMP thermal compliance with Eupry
Get an instant overview of how all the compliance solution works, the technical specifications, and much more.
Frequently asked questions about GMP temperature compliance
GMP references and regulatory sources
EU GMP guidelines:
- EudraLex Volume 4, Annex 15: Qualification and Validation (2015) Read here
- EudraLex Volume 4, Annex 11: Computerised Systems (2011), with draft revision July 2025 Read here
US FDA guidance:
- 21 CFR Part 210: Current Good Manufacturing Practice in Manufacturing, Processing, Packing, or Holding of Drugs Read here
- 21 CFR Part 211: Current Good Manufacturing Practice for Finished Pharmaceuticals Read here
- 21 CFR Part 11: Electronic Records; Electronic Signatures (1997) Read here
International standards:
- WHO Technical Report Series No. 961, Annex 9: Model guidance for the storage and transport of time- and temperature-sensitive pharmaceutical products (2011) Read here
- ICH Q9(R1): Quality Risk Management Read here
- ICH Q10: Pharmaceutical Quality System Read here
- ISPE Good Practice Guide: Controlled Temperature Chambers (2nd edition, 2021)
- ISO 17025: General requirements for the competence of testing and calibration laboratories
Data integrity guidance:
Learn more

Guidelines for Good Distribution Practice (GDP)
Good Distribution Practice (GDP) sets the standard for safe pharmaceutical logistics. Understand the requirements, why they matter, and how to stay compliant.
Thermal compliance solutions for GMP
Automated monitoring in real-time, validation services, and the fastest calibration you will find: One vendor, one solution, everything you need for thermal compliance in GMP.

Key regulations of temperature compliance
From FDA 21 CFR part 11 to ISO 17025 and ISO 9011 - see the complete guide to the guidelines, standards, and regulations that apply to thermal compliance in GxP.