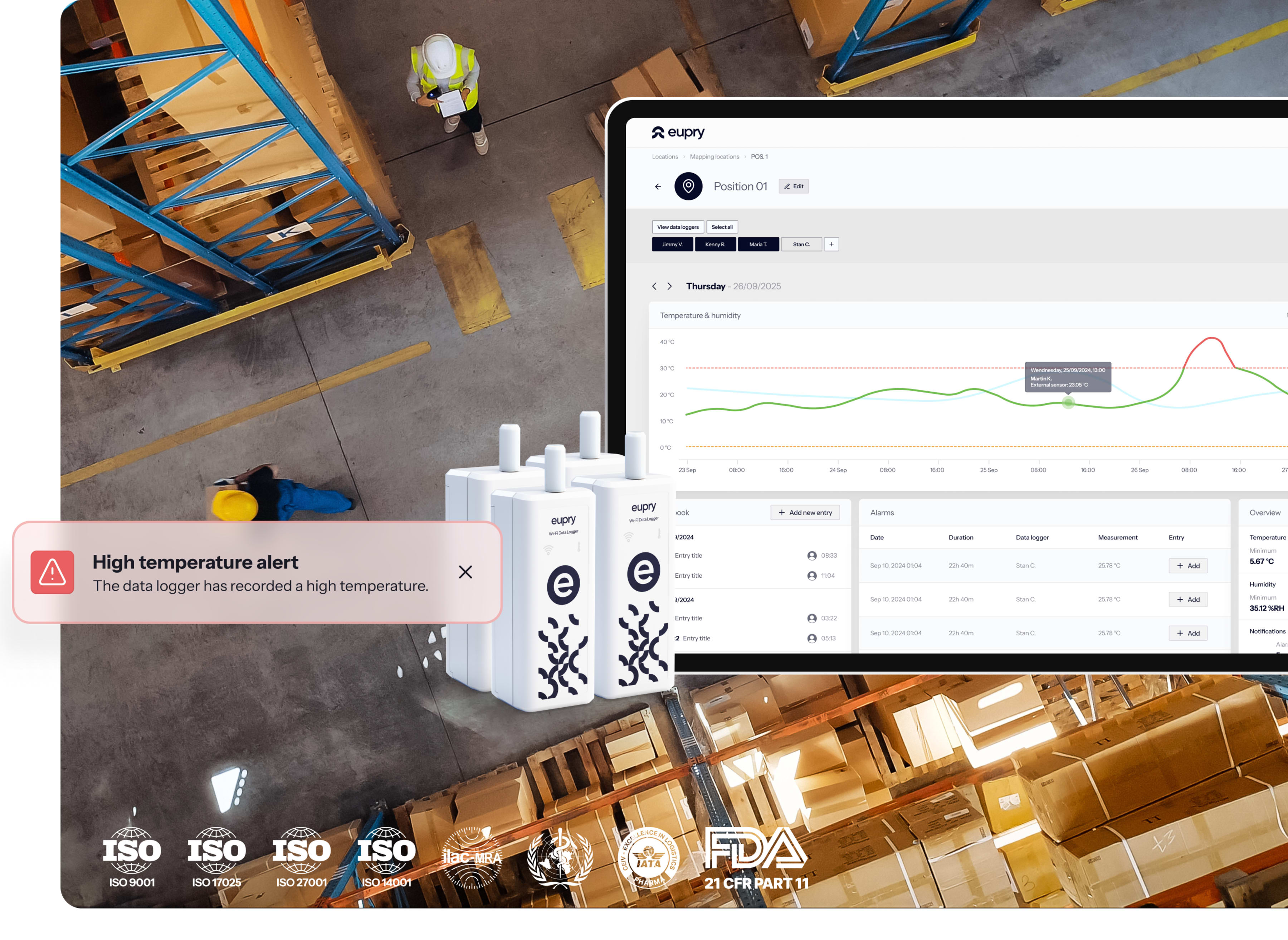
Warehouse monitoring for pharmaceutical logistics
GDP compliance documentation that impresses clients. Validation that does not disrupt operations. One provider for monitoring, mapping, and calibration.
Too many vendors, too slow, too manual
Pharmaceutical warehouses face complex monitoring requirements across loading docks, storage zones, and controlled areas. Traditional setups require multiple vendors for monitoring, mapping, and calibration. Winter and summer mapping cycles create validation bottlenecks that delay facility readiness and revenue.

Faster validation, one provider, one platform
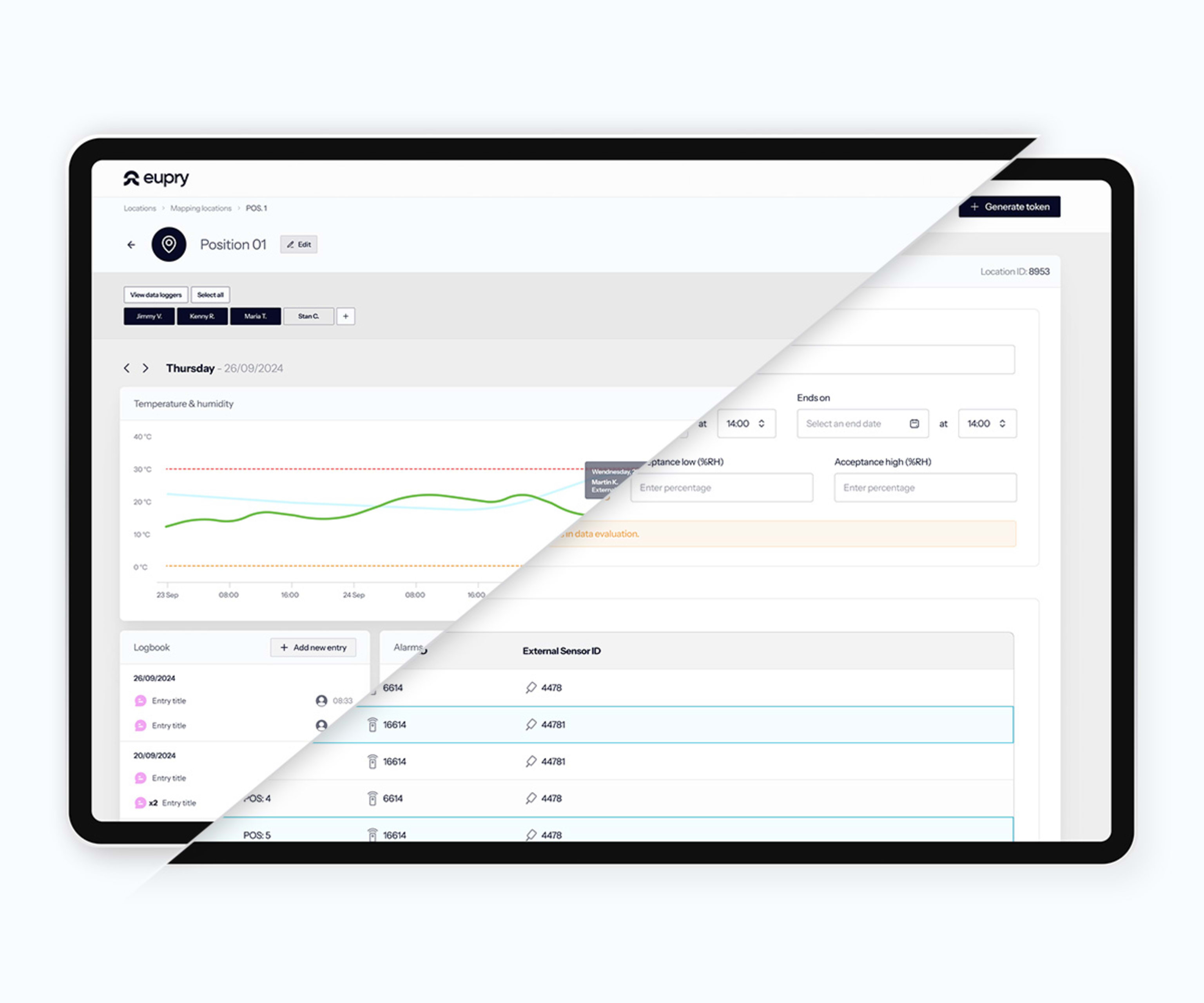
Eupry delivers complete warehouse GxP compliance from one provider. Wireless mapping and monitoring covers all zones with automated alerts and real-time visibility. Digital mapping validates faster with less downtime. On-site accredited calibration happens in seconds. One platform, single source of truth, globally.


“Eupry was very professional and informative throughout the process, providing us with high-quality service (...) It's undoubtedly a well-put partnership.”
Joakim Sund
Quality Assurance Specialist

“Eupry was instrumental in helping us establish a GDP-compliant operation ready for the standards of pharmaceutical logistics. Their flexibility, expertise, and collaborative approach were exactly what we needed to navigate the requirements of the industry and develop a foundation that worked for us."

Michael O'Brien
President

Great service! The Eupry Team listens and provides a rapid response. That, added to their top-notch technology, provides accurate mapping and monitoring within industry rules and regulations and peace of mind.

Yolanda Rodriguez
Executive Operations Administrator
Relevant products

Download a product catalog
Get an instant overview of every sensor, spec, and product option for monitoring and mapping – in one PDF.
Want to know more?
Discover how Eupry's automated compliance (monitoring, calibration, and validation) solutions work today.