Temperature compliance for healthcare logistics
Reduce risks, manual work, and costs with reliable, harmonized temperature monitoring, mapping, and calibration designed for the diverse needs of healthcare logistics.
- GDP compliance in weeks
- Audit reports in 3 clicks
- Standardized processes across sites
- 20-30% reduced costs


You can't win pharma without GDP compliance
... but getting there and that is both complex and time-consuming.
Dozens of vendors (and problems)
Validation consultants. Monitoring systems. Calibration services. Each with different processes, timelines, and bills. Managing multiple vendors is complex and costly.
Validations delay expansion and growth
Every warehouse layout change means re-mapping. Every new facility sits idle waiting for final validation. Your expansion plans wait on other's schedules.
Quality teams spread too thin
QA firefight across sites instead of building systems. Local teams are disturbed. Local problems become global headaches. Compliance is reactive, not strategic.
Costs that spiral without warning
Surprise calibration costs. Emergency re-mapping fees. Last-minute consultant rates. Growth is expensive when every expansion restarts the cycle and vendors are scattered.
One vendor, all you need for thermal compliance in GDP
Pharma clients are among the top most valuable segments for logistics, but they also come with the strictest standards.
Our system was built for this complexity.
We provide the expertise and services to create a solid foundation on which healthcare logistics providers can expand their pharma operation - faster, cheaper, and with no downtime.
CEIV & GDP ready
The solutuon is built for GDP and CEIV, and both mapping and monitoring requirements satisfied.
20-30% lower costs
20-30% lower total cost of ownership over 3-5 years. Automate and go from 10, 20, 30, or 100+ vendors to 1.
Higher quality levels
2-6x richer data for better decisions, predictive mainentance, and proactive quality management.
Central control of all sites
Same SOPs, same dashboards, same documentation for all facilities and unit across your network.
Trusted by 1000+ GxP companies
Monitoring, continuous mapping, and calibration in ONE
Bring temperature and humidity compliance – monitoring, calibration, and mapping – into one digital, GxP-compliant solution, providing one centralized overview of all data and one process.
- Reduce total cost of ownership
- Eliminate waste of time spent on manual tasks
- Gain full control (and GDP-compliance) across facilities

Temperature mapping services: Fast validation + continuous compliance
Get your facilities GDP-ready in weeks.
Engineer-led temperature mapping validates your warehouses or other storage facilities quickly, and then continuous mapping eliminates periodic re-mappings.
- Fast GDP validation: Fast project delivery and flexible solutions.
- Digital mapping: Digital test plans and reporting from anywhere.
- Lower costs: 20-30% lower total cost of ownership.
- Higher quality: Well-tested processes, audit support, and more.

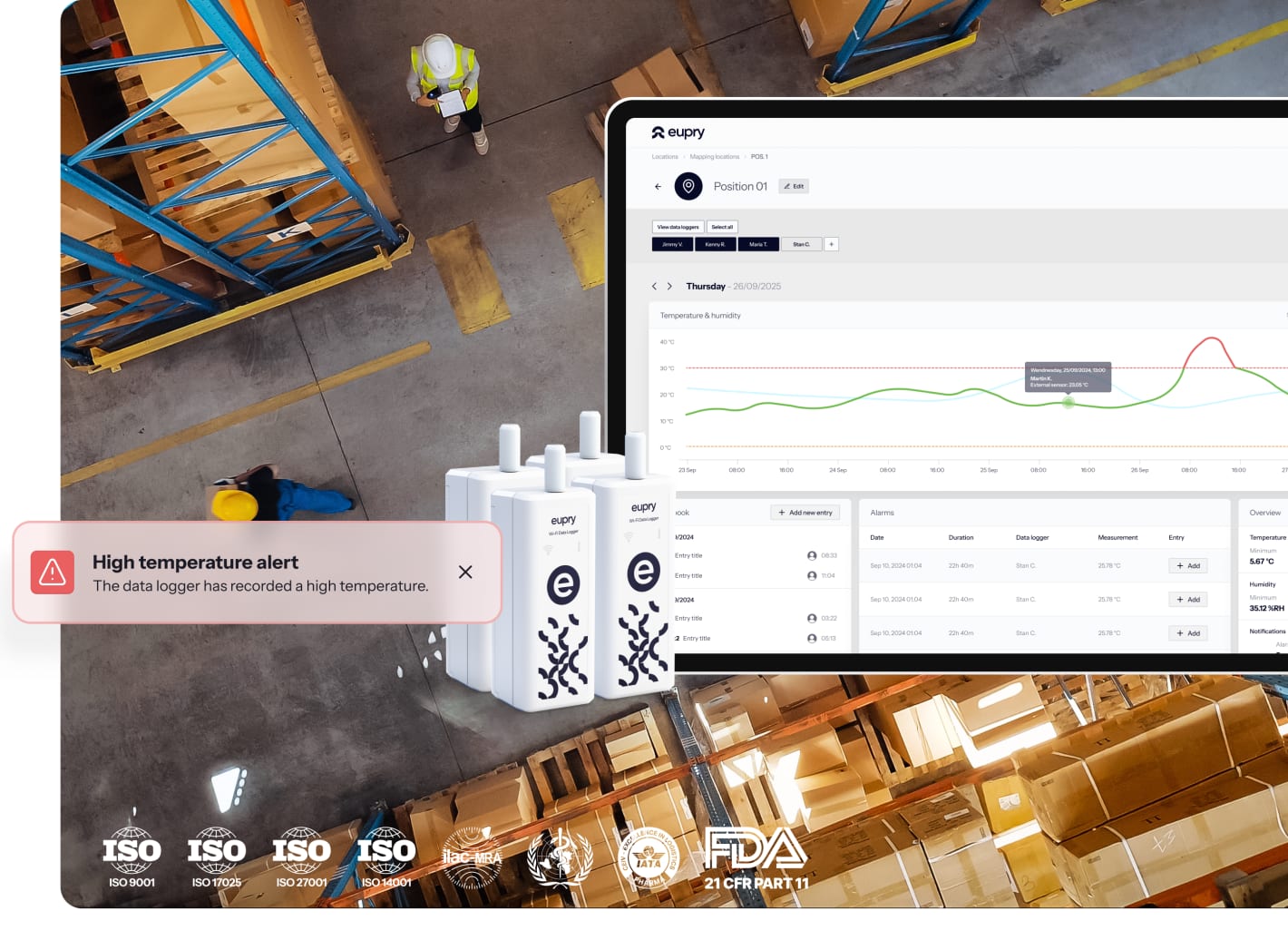
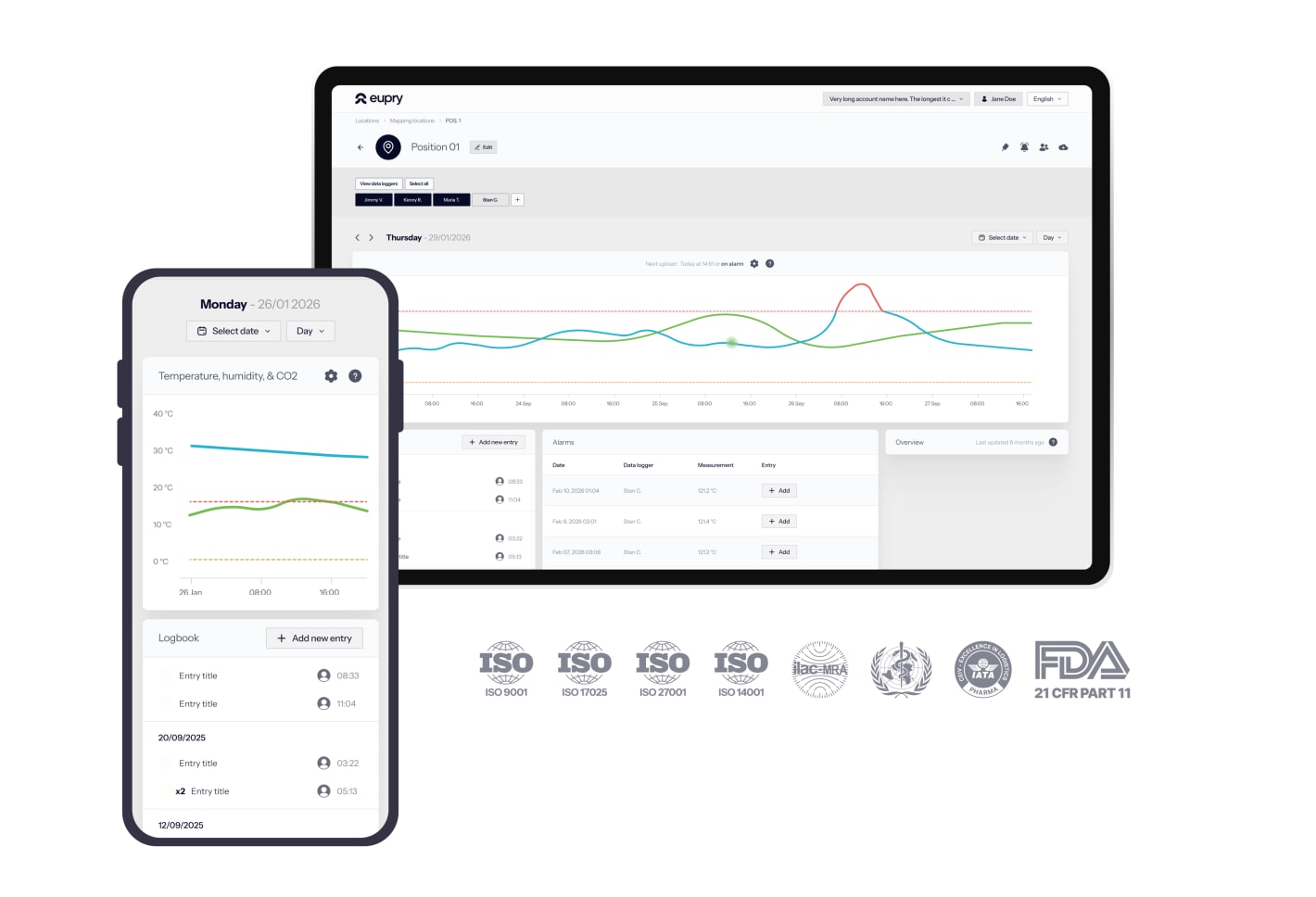
Live automated monitoring = central control across all sites
Wi-Fi-based monitoring provides one centralized view of temperature data across your entire logistics network.
- See every facility on one dashboard
- CEIV and GDP ready by design
- Audit reports in 3 clicks
- Proactive alerts via email/SMS
Accredited on-the-wall calibration: 95% faster, zero downtime
For a large logistics facility, traditional calibration might take days; Eupry's ISO 17025-accredited on-the-wall calibration takes under an hour.
Swap sensor tips in seconds without removing loggers, and reduce calibration time by up to 95%.
No device removal, no downtime, no data gaps.
Get an instant overview of how the solutions work.

Download a free product catalog
See all the technical details and learn how the GDP-ready temperature compliance solutions work.
From 15, 40, 100+ vendors to 1
Most operations juggle separate vendors for facility monitoring, shipment trackers, validation services, calibration labs, and more.
With Eupry, you get one integrated solution.
- One vendor relationship, contract, and support team
- One compliance framework across networks
- Predictable - and reduced - compliance budget


Great service! The Eupry Team listens and provides a rapid response. That, added to their top-notch technology, provides accurate mapping and monitoring within industry rules and regulations and peace of mind.

Yolanda Rodriguez
Executive Operations Administrator

“Eupry was very professional and informative throughout the process, providing us with high-quality service (...) It's undoubtedly a well-put partnership.”
Joakim Sund
Quality Assurance Specialist

“I have nothing but praise for Eupry. They have invented a system that is simple and does everything it promises. Loggers are reliable and the user interface is logical, intuitive, and detailed in a way so I can easily monitor and pull out reports.”

Allan Witt
GDP Responsible Person at Worldwide Flight Service
What makes Eupry the right fit for pharma logistics?
Healthcare logistics providers use Eupry's services to prove GDP compliance quickly, cut vendors, and reduce costs.
- Validation expertise that gets logistics: Our team has qualified hundreds of pharma facilities and know what is required.
- Technology designed for multi-site networks: One dashboard for all sites, standard global processes, and fast, scalable deployment.
- Integrated platform = no vendor chaos: Instead of juggling vendors for mapping, monitoring, and calibration, you get one partner who handles everything.
- Speed that wins contracts: GDP-ready in months means you can bid on contracts while competitors are still validating their first site.
- No more re-mapping: Our continuous mapping solution eliminates disruptive re-mappings. Maintain compliance without shutting down your facilities.

Built for GDP
All your compliance boxes? Consider them ticked.
- ISO 9001 certified
- ISO 17025 accredited
- ISO 27001 accredited
- ISO 14001 certified
- 21 CFR Part 11 module
And much more.

Devices for all stages
The cold chain monitoring system uses two sensor types designed for:
Transit: Cellular smart labels stream temperature, GPS, shock, humidity, and light in real time.
Storage: Wi-Fi-based wireless data loggers continuously monitor conditions in facilities.
What they have in common:
- Live data and alerts
- Complete audit trail
- Global coverage

Legal basis and guidance
The solutions are designed for GDP with a legal basis in cGMP, USP <1079>, and EudraLex Volume 4 Part 1 Chapter 3, and based on guidance from WHO guidelines (supplement 8 and 7 and annex 8), DKD-R 5, FD X 15-140 (French standard), and the ISPE Standard “Controlled Temperature Chamber Mapping and Monitoring”.
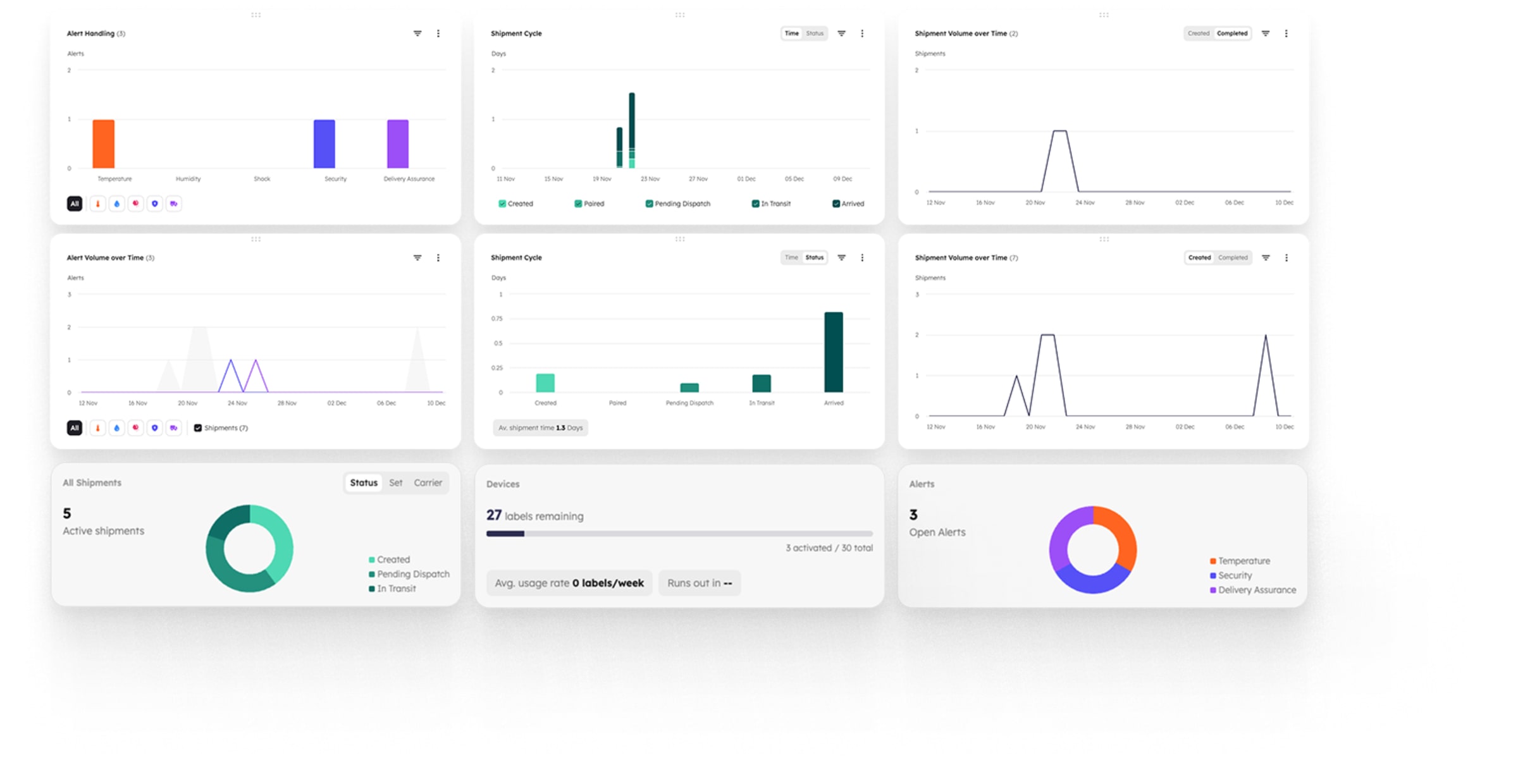
One system, all your cold chain data
No more switching between systems.
Get a single source of truth of all products from storage through transport to delivery.
- Protect products with live data: Instant alerts let you intervene.
- Automate workflow: No manual work with automated data.
- AI analytics: Detects anomalies and forecasts delays.
CEIV Pharma ready - faster
Eupry's solution is designed for GDP and supports all requirements for CEIV Pharma certification.
- CEIV documentation: The platform provides all the temperature documentation that CEIV auditors expect.
- Faster certification: Logistics operators using Eupry can typically get CEIV Pharma certified in 6 months compared to the normal 12-18 months.
- All the support you need: Our team can help prepare documentation and support you through the certification process.
Get in touch with one of our GDP specialists for a no-strings-attached consultation.

How DSV validated their GDP-compliant logistics center with Eupry
The response times and availability from Eupry were very impressive. They helped us carry out various tests and assisted in the interpretation of the data (...) It was undoubtedly a well-put partnership.
Joakim Sund, Quality Assurance Specialist at DSV

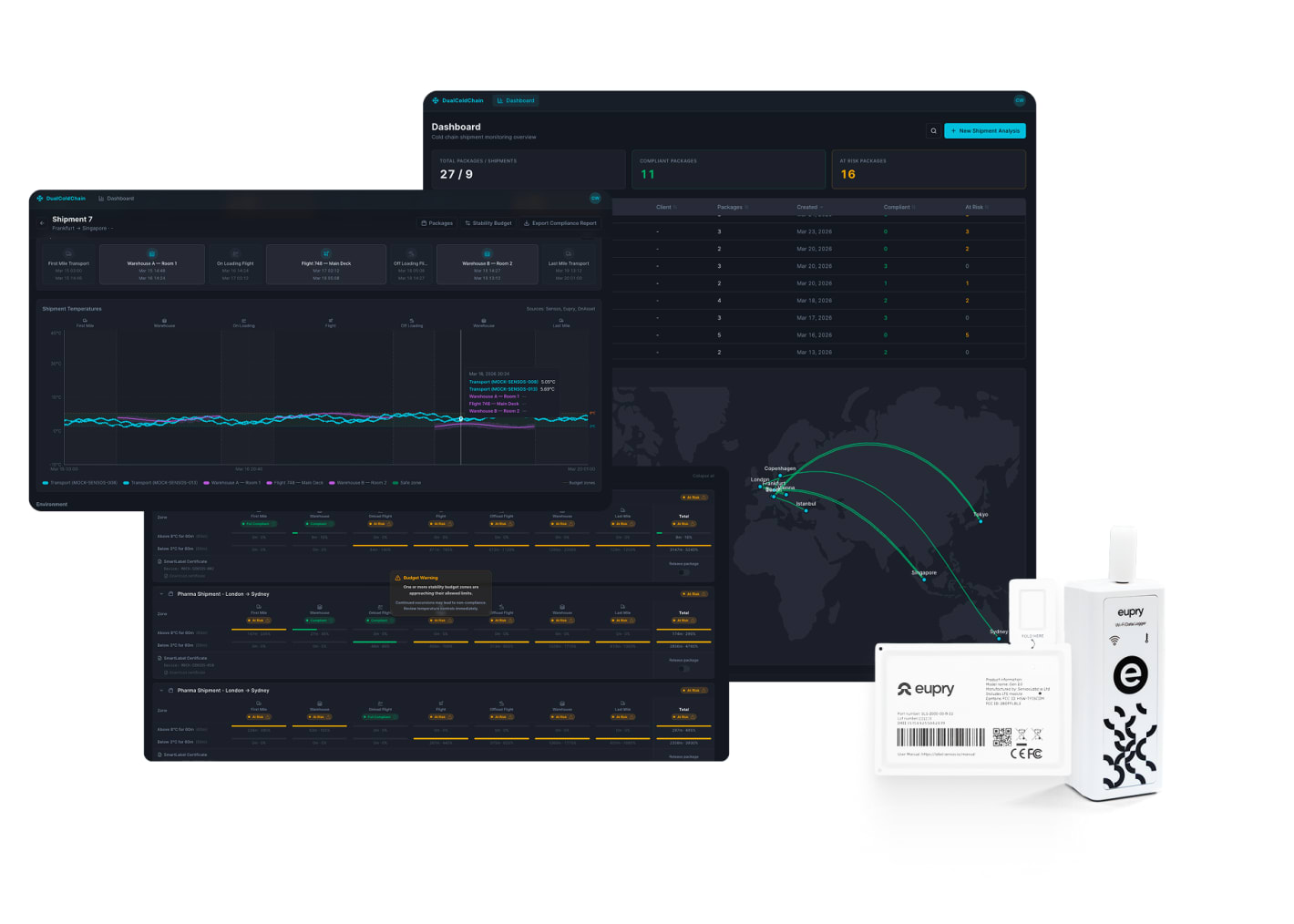
No product loss with full cold chain control
– from warehouse to delivery
Know instantly when issues arise anywhere in the cold chain with the Dual Compliance Platform.
- Track stability budget live across the full journey
- Adjust handling to protect products
- Get one audit trail from storage to destination
- Shipment and facility tracking in one
High compliance rating by Novo Nordisk
Eupry Aps provided (…) documentation to conclude that procedures and efficient controls to ensure compliance are in place and well-functioning as intended in all key areas/processes (…)
ISO AUDIT REPORT by Novo Nordisk, June 2023


Send us a message
Need more information or a tailored price? Fill out the form, and we will be in touch asap!





