Eupry for the pharma industry
One provider for monitoring, calibration, mapping, and validation
- Built and validated for GMP and GDP requirements
- Temperature, humidity, CO2, and differential pressure
- Complete audit trails in clicks – from calibration to reports
Monitoring and validation gaps are among the most common audit findings in pharma
- and most compliance setups make them inevitable
Fragmented systems, fragmented data
Temperature, humidity, differential pressure, and CO2 records, calibration certificates, reports, and more spread across systems, making audits risky and time-consuming.
Validation backlogs - and disruptions
IQ/OQ delays hold up facilities, re-mappings pile up. Your team is stuck choosing between operations and clearing the qualification queue.
Manual processes that risk traceability and waste time
Manual data transfers, deviation logs, and reporting. Every manual task is a place where traceability can break and you could spend time on something else.
More vendors, more costs, more coordination
One provider for monitoring, another for calibration, a third for mapping: three contracts, timelines, costs, and points of contact - and that is just for one site.
One vendor, all you need for environmental compliance in pharma
Monitoring, calibration, mapping, and validation in one solution with one vendor. One audit trail, one data format, one point of contact.

Trusted by more than 1000+ organizations worldwide

Validation
GxP-tailored mapping equipment, software, and services let you run faster, more reliable mappings.
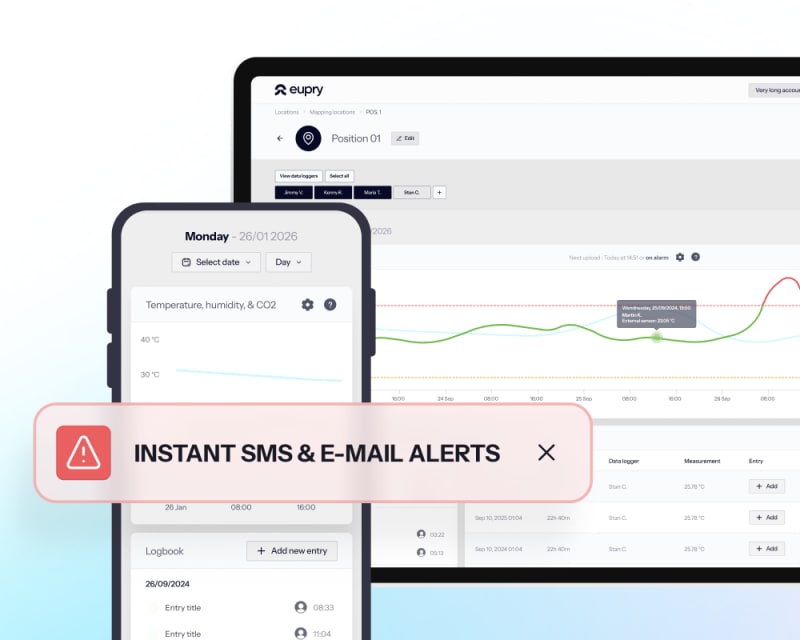
Monitoring
Automated Wi-Fi-based monitoring with proactive alerts and audit-ready reports across sites and units.

Calibration
ISO 17025-accredited, automated calibration that reduces calibration time spend by up to 95%.

Download a product catalog
Get an instant overview of all the technical details, solution options, and how everything works.
Designed for pharmaceutical requirements
All your compliance boxes? Consider them ticked.
- ISO 17025-accredited calibration
- ISO 9001-certified quality
- ISO 27001‑certified data security
- ISO 14001-certified environmental management.
The solution is fully validated and built for FDA 21 CFR Part 11, EU GMP Annex 11 and Annex 15, and GDP requirements.

High compliance rating by Novo Nordisk
Eupry Aps provided (…) documentation to conclude that procedures and efficient controls to ensure compliance are in place and well-functioning as intended in all key areas/processes.
ISO AUDIT REPORT by Novo Nordisk, June 2023

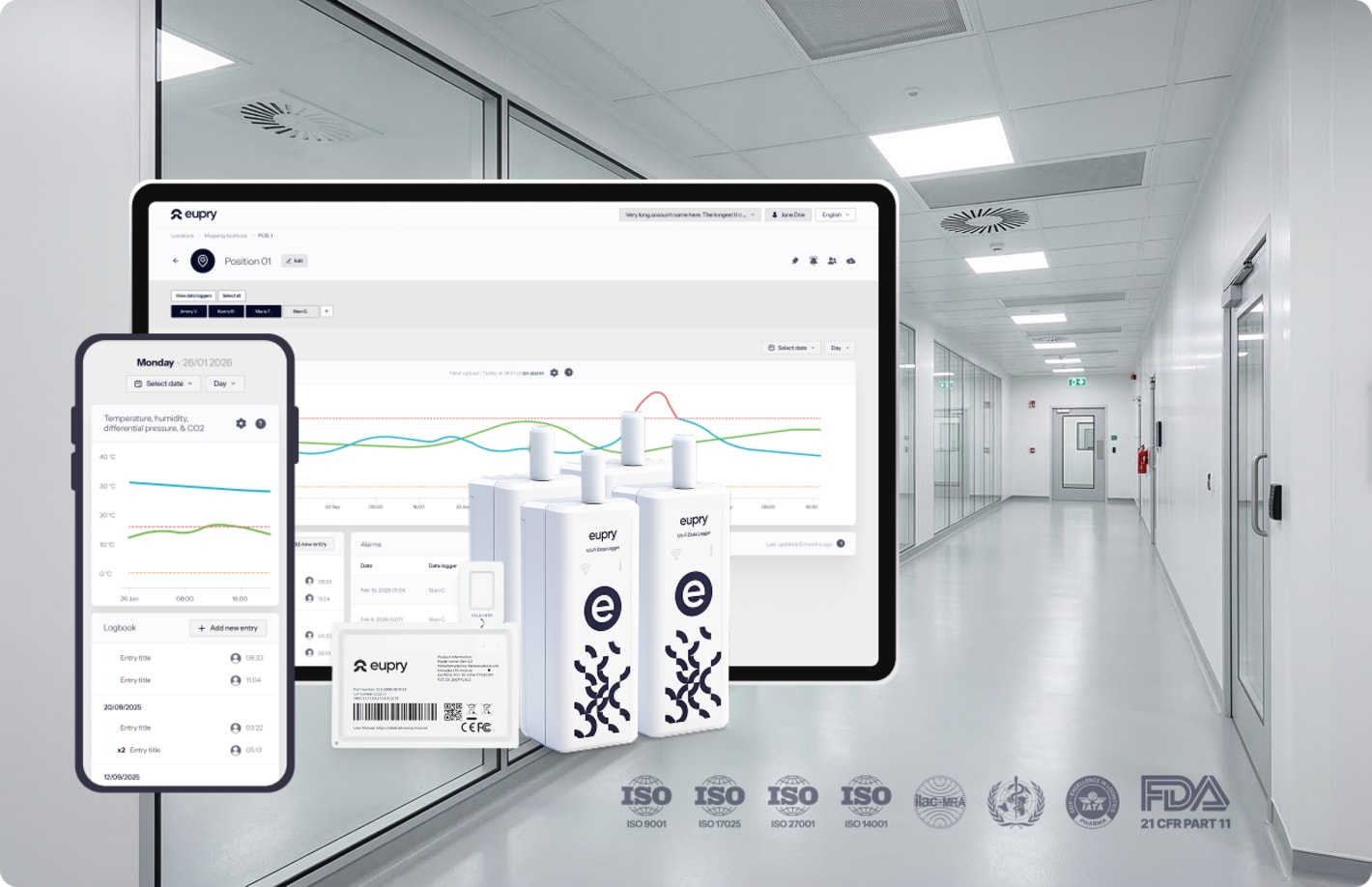
Multi-parameter monitoring - with full central overview
Get a single compliance overview across all parameters, for all facilities, departments, and units, in one validated platform.
- Temperature, humidity, CO2, and differential pressure
- Live overview on any screen
- Instant deviation alerts
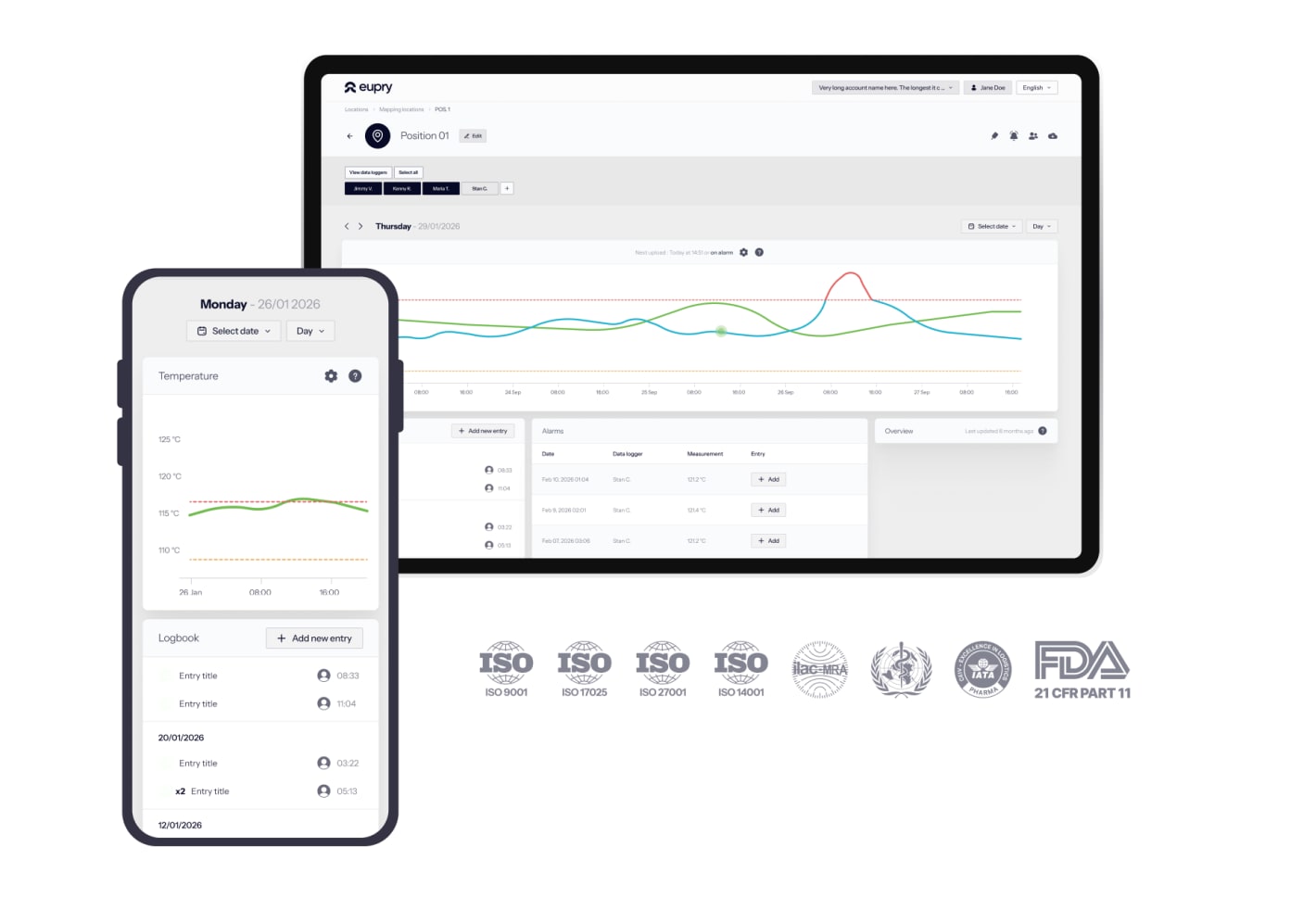
Manual work turned automated
With Eupry's wireless data loggers and platform, data transfer, deviation logging, and audit reporting is all automated.
Plus, our patented and ISO 17025-accredited sensor-tip calibration lets you calibrate on location in seconds.

On-site calibration without downtime
Old sensor out. New sensor in. Calibration is done.
Eupry’s patented solution redefines how calibration is done in GxP, letting you handle all your calibrations in minutes, directly at site, without changing data loggers.
= 95% less time spent on calibration.
- Calibration handled in-house
- Monitoring stays active
- Certificates are automatically stored
Mapping and validation services with minimal downtime
From rentable mapping kits to fully managed on-site studies, all our validation services are all designed to meet pharma requirements.
- Digital validation: Create protocols, reports, and track the study live in the platform.
- Focus on your operation: Let our experienced specialists handle some or all of your validation.
- No downtime: Fast, continuous setup keep operational disruptions to a minimum.

What our clients say
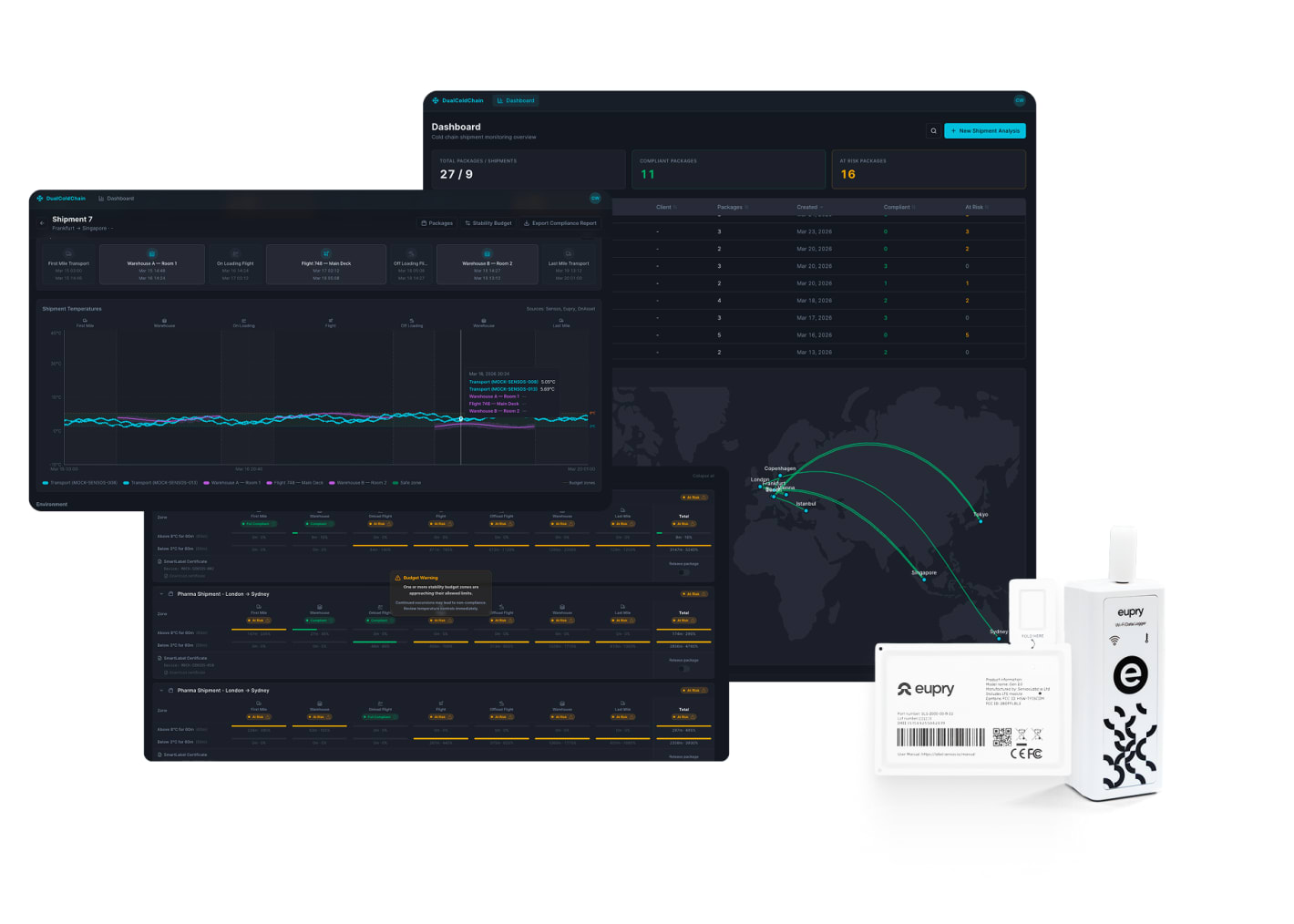
No product loss with full cold chain control
– from warehouse to delivery
Pharmaceutical companies lose an estimated $35 billion annually to cold chain failures. With Eupry, you can act instantly when an issue arises anywhere in the journey with end-to-end cold chain control.
- Track stability budget live across the full journey
- Adjust handling to protect products
- Get one audit trail from storage to destination
- Shipment and facility tracking in one
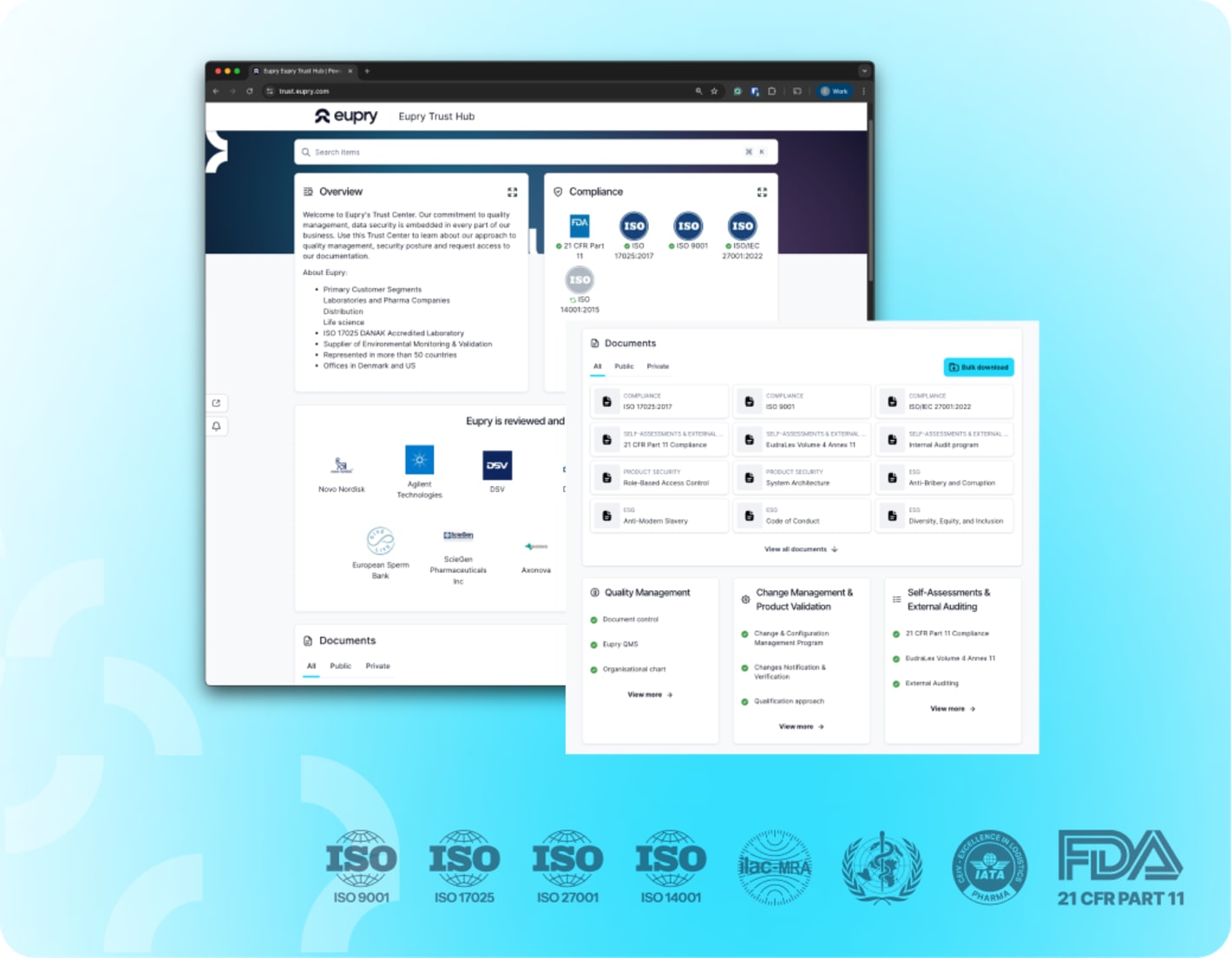
The Eupry Trust Hub
All compliance documentation in one place
Vetting a new partner involves a lot of questions, especially in pharma. Our Trust Hub is designed to make that process easier. It gathers all the information you need to assess our compliance and processes – from accreditations, certifications, and quality management approach to how we handle data security.

Send us a message
Need more information or a tailored price? Fill out the form, and we will be in touch asap!

Eupry's system is easy to use, and they offer great support.
Anette Fritzon
CQA Warehouse Officer

“The calibration of the Eupry temperature loggers is easy and quick. I received a box with the new sensors (…) It took me under an hour to calibrate 40 loggers – including the time it took to find the refrigerators.”

Kasper H. Christophersen
Research Associate at Novozymes

"We have saved around 50-70% of the time we spent on temperature monitoring."

Anders Rasmussen
Senior Logistic Manager at Dechra Pharmaceuticals PLC
Use cases
From cold storage rooms to autoclaves, Eupry's solutions cover thermal compliance for every pharmaceutical environment.
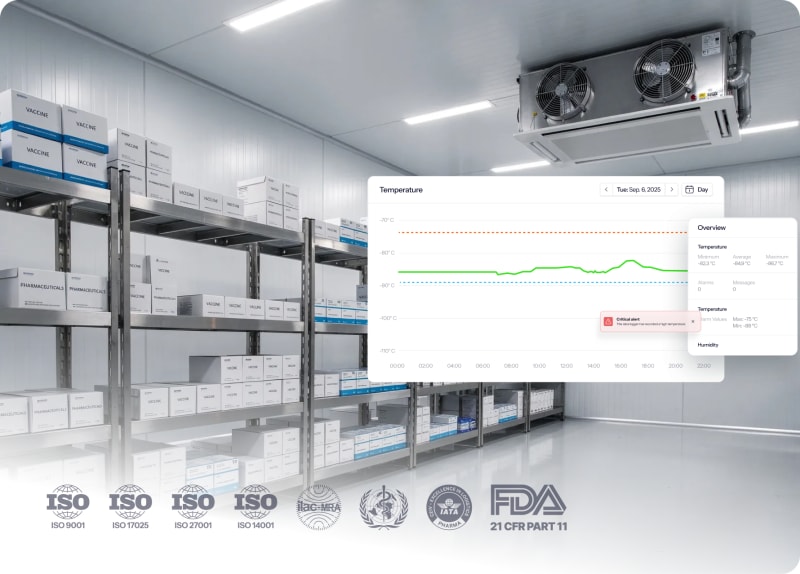
Cold storage rooms
Complete visibility across multiple cold rooms and sites. Standardized processes and audit-ready documentation for FDA, EMA, and more.

Fridges and freezers
24/7 visibility for vaccines, biologics, and temperature-sensitive samples. Instant alerts, built-in redundancy, and audit-ready documentation for FDA, EMA, and more.

Incubators and climate chambers
You run stability studies or cell cultures that require precise and stable temperature and humidity control. You need reliable monitoring that catches problems immediately, not during the next check.

Laboratories
You run a lab with multiple assets requiring environmental control with more than just temperature. You need one system that monitors everything and handles compliance automatically.

Ultra low freezers (ULTs) and cryos
24/7 visibility for samples stored from -80°C to -196°C. Instant alerts, built-in redundancy, and audit-ready documentation for FDA, EMA, and more.

Warehouses
GDP compliance documentation that impresses clients. Validation that does not disrupt operations. One provider for monitoring, mapping, and calibration.
Other industries: Biotech · CDMOs · Healthcare logistics
Get started
Get the product catalog for technical specifications, solution options, and pricing details. Or send us a message – we typically respond within a few hours.





